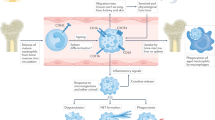
Key Points
-
Neutrophils contribute to rheumatoid arthritis (RA) pathology through the release of cytotoxic and immunoregulatory molecules
-
Neutrophils from patients with RA have an activated phenotype: delayed apoptosis, increased capacity to produce reactive oxygen species, active gene expression and membrane expression of high-affinity Fcγ receptors
-
In mouse models of RA, neutrophils are critical to initiation and progression of the disease
-
Neutrophil extracellular traps might be a source of citrullinated autoantigens (hypercitrullinated cytoplasmic proteins and citrullinated histones) in RA
-
Anticitrullinated protein antibodies are a hallmark of RA, and high titres of such autoantibodies are associated with severe disease
Abstract
Of all cells implicated in the pathology of rheumatoid arthritis (RA), neutrophils possess the greatest cytotoxic potential, owing to their ability to release degradative enzymes and reactive oxygen species. Neutrophils also contribute to the cytokine and chemokine cascades that accompany inflammation, and regulate immune responses via cell–cell interactions. Emerging evidence suggests that neutrophils also have a previously unrecognised role in autoimmune diseases: neutrophils can release neutrophil extracellular traps (NETs) containing chromatin associated with granule enzymes, which not only kill extracellular microorganisms but also provide a source of autoantigens. For example, citrullinated proteins that can act as neoepitopes in loss of immune tolerance are generated by peptidylarginine deiminases, which replace arginine with citrulline residues, within neutrophils. Indeed, antibodies to citrullinated proteins can be detected before the onset of symptoms in patients with RA, and are predictive of erosive disease. Neutrophils from patients with RA have an increased tendency to form NETs containing citrullinated proteins, and sera from such patients contain autoantibodies that recognize these proteins. Thus, in addition to their cytotoxic and immunoregulatory role in RA, neutrophils may be a source of the autoantigens that drive the autoimmune processes underlying this disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Viatte, S., Plant, D. & Raychaudhuri, S. Genetics and epigenetics of rheumatoid arthritis. Nat. Rev. Rheumatol. 9, 141–153 (2013).
Reynisdottir, G. et al. Structural lung changes and local anti-citrulline immunity are early features of anti citrullinated-proteins antibodies positive rheumatoid arthritis. Arthritis Rheum. http://dx.doi.org/10.1002/art.38201.
Quirke, A. M. et al. Heightened immune response to autocitrullinated Porphyromonas gingivalis peptidylarginine deiminase: a potential mechanism for breaching immunologic tolerance in rheumatoid arthritis. Ann. Rheum. Dis. 73, 263–269 (2014).
Maresz, K. J. et al. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathog. 9, e1003627 (2013).
Sabeh, F., Fox, D. & Weiss, S. J. Membrane-type I matrix metalloproteinase-dependent regulation of rheumatoid arthritis synoviocyte function. J. Immunol. 184, 6396–6406 (2010).
Miller, M. C. et al. Membrane type 1 matrix metalloproteinase is a crucial promoter of synovial invasion in human rheumatoid arthritis. Arthritis Rheum. 60, 686–697 (2009).
Tolboom, T. C. et al. Invasive properties of fibroblast-like synoviocytes: correlation with growth characteristics and expression of MMP-1, MMP-3, and MMP-10. Ann. Rheum. Dis. 61, 975–980 (2002).
Murphy, G. & Nagase, H. Reappraising metalloproteinases in rheumatoid arthritis and osteoarthritis: destruction or repair? Nat. Clin. Pract. Rheumatol. 4, 128–135 (2008).
Smolen, J. E. et al. L-selectin signaling of neutrophil adhesion and degranulation involves p38 mitogen-activated protein kinase. J. Biol. Chem. 275, 15876–15884 (2000).
Yang, L. et al. ICAM-1 regulates neutrophil adhesion and transcellular migration of TNF-α-activated vascular endothelium under flow. Blood 106, 584–592 (2005).
Woodfin, A., Voisin, M. B. & Nourshargh, S. Recent developments and complexities in neutrophil transmigration. Curr. Opin. Hematol. 17, 9–17 (2010).
Cross, A., Barnes, T., Bucknall, R. C., Edwards, S. W. & Moots, R. J. Neutrophil apoptosis in rheumatoid arthritis is regulated by local oxygen tensions within joints. J. Leukoc. Biol. 80, 521–528 (2006).
Walmsley, S. R. et al. Hypoxia-induced neutrophil survival is mediated by HIF-1α-dependent NF-κB activity. J. Exp. Med. 201, 105–115 (2005).
Wright, H. L., Thomas, H. B., Moots, R. J. & Edwards, S. W. RNA-seq reveals activation of both common and cytokine-specific pathways following neutrophil priming. PLoS ONE 8, e58598 (2013).
Cassatella, M. A. Neutrophil-derived proteins: selling cytokines by the pound. Adv. Immunol. 73, 369–509 (1999).
Mantovani, A., Cassatella, M. A., Costantini, C. & Jaillon, S. Neutrophils in the activation and regulation of innate and adaptive immunity. Nat. Rev. Immunol. 11, 519–531 (2011).
Fujishima, S. et al. Regulation of neutrophil interleukin 8 gene expression and protein secretion by LPS, TNF-α, and IL-1β. J. Cell Physiol. 154, 478–485 (1993).
Nunes, P., Demaurex, N. & Dinauer, M. C. Regulation of the NADPH oxidase and associated ion fluxes during phagocytosis. Traffic 14, 1118–1131 (2013).
Fossati, G., Bucknall, R. C. & Edwards, S. W. Insoluble and soluble immune complexes activate neutrophils by distinct activation mechanisms: changes in functional responses induced by priming with cytokines. Ann. Rheum. Dis. 61, 13–19 (2002).
Fossati, G., Bucknall, R. C. & Edwards, S. W. Fcγ receptors in autoimmune diseases. Eur. J. Clin. Invest. 31, 821–831 (2001).
Robinson, J., Watson, F., Bucknall, R. C. & Edwards, S. W. Activation of neutrophil reactive-oxidant production by synovial fluid from patients with inflammatory joint disease. Soluble and insoluble immunoglobulin aggregates activate different pathways in primed and unprimed cells. Biochem. J. 286 (Pt 2), 345–351 (1992).
Stockley, R. A. Neutrophils and the pathogenesis of COPD. Chest 121, 151S–155S (2002).
Derouet, M., Thomas, L., Cross, A., Moots, R. J. & Edwards, S. W. Granulocyte macrophage colony-stimulating factor signaling and proteasome inhibition delay neutrophil apoptosis by increasing the stability of Mcl-1. J. Biol. Chem. 279, 26915–26921 (2004).
Derouet, M. et al. Sodium salicylate promotes neutrophil apoptosis by stimulating caspase-dependent turnover of Mcl-1. J. Immunol. 176, 957–965 (2006).
Thomas, L. W., Lam, C. & Edwards, S. W. Mcl-1; the molecular regulation of protein function. FEBS Lett. 584, 2981–2989 (2010).
Savill, J. S. et al. Macrophage phagocytosis of aging neutrophils in inflammation. Programmed cell death in the neutrophil leads to its recognition by macrophages. J. Clin. Invest. 83, 865–875 (1989).
Eggleton, P., Wang, L., Penhallow, J., Crawford, N. & Brown, K. A. Differences in oxidative response of subpopulations of neutrophils from healthy subjects and patients with rheumatoid arthritis. Ann. Rheum. Dis. 54, 916–923 (1995).
Wright, H. L., Chikura, B., Bucknall, R. C., Moots, R. J. & Edwards, S. W. Changes in expression of membrane TNF, NF-κB activation and neutrophil apoptosis during active and resolved inflammation. Ann. Rheum. Dis. 70, 537–543 (2011).
Matsumoto, T. et al. The membrane proteinase 3 expression on neutrophils was downregulated after treatment with infliximab in patients with rheumatoid arthritis. Clin. Appl. Thromb. Hemost. 14, 186–192 (2008).
Wittkowski, H. et al. Effects of intra-articular corticosteroids and anti-TNF therapy on neutrophil activation in rheumatoid arthritis. Ann. Rheum. Dis. 66, 1020–1025 (2007).
Chakravarti, A., Raquil, M. A., Tessier, P. & Poubelle, P. E. Surface RANKL of Toll-like receptor 4-stimulated human neutrophils activates osteoclastic bone resorption. Blood 114, 1633–1644 (2009).
Assi, L. K. et al. Tumor necrosis factor α activates release of B lymphocyte stimulator by neutrophils infiltrating the rheumatoid joint. Arthritis Rheum. 56, 1776–1786 (2007).
Cross, A., Bucknall, R. C., Cassatella, M. A., Edwards, S. W. & Moots, R. J. Synovial fluid neutrophils transcribe and express class II major histocompatibility complex molecules in rheumatoid arthritis. Arthritis Rheum. 48, 2796–2806 (2003).
Lally, F. et al. A novel mechanism of neutrophil recruitment in a coculture model of the rheumatoid synovium. Arthritis Rheum. 52, 3460–3469 (2005).
Parsonage, G. et al. Prolonged, granulocyte-macrophage colony-stimulating factor-dependent, neutrophil survival following rheumatoid synovial fibroblast activation by IL-17 and TNFα. Arthritis Res. Ther. 10, R47–R59 (2008).
Raza, K. et al. Synovial fluid leukocyte apoptosis is inhibited in patients with very early rheumatoid arthritis. Arthritis Res. Ther. 8, R120–R127 (2006).
Weinmann, P. et al. Delayed neutrophil apoptosis in very early rheumatoid arthritis patients is abrogated by methotrexate therapy. Clin. Exp. Rheumatol. 25, 885–887 (2007).
Rollet-Labelle, E. et al. Cross-linking of IgGs bound on circulating neutrophils leads to an activation of endothelial cells: possible role of rheumatoid factor in rheumatoid arthritis-associated vascular dysfunction. J. Inflamm. (Lond.) 10, 27 (2013).
Nurcombe, H. L., Bucknall, R. C. & Edwards, S. W. Neutrophils isolated from the synovial fluid of patients with rheumatoid arthritis: priming and activation in vivo. Ann. Rheum. Dis. 50, 147–153 (1991).
Watson, F., Robinson, J. J., Phelan, M., Bucknall, R. C. & Edwards, S. W. Receptor expression in synovial fluid neutrophils from patients with rheumatoid arthritis. Ann. Rheum. Dis. 52, 354–359 (1993).
Quayle, J. A., Watson, F., Bucknall, R. C. & Edwards, S. W. Neutrophils from the synovial fluid of patients with rheumatoid arthritis express the high affinity immunoglobulin G receptor, FcγRI (CD64): role of immune complexes and cytokines in induction of receptor expression. Immunology 91, 266–273 (1997).
Robinson, J. J., Watson, F., Bucknall, R. C. & Edwards, S. W. Role of Fcγ receptors in the activation of neutrophils by soluble and insoluble immunoglobulin aggregates isolated from the synovial fluid of patients with rheumatoid arthritis. Ann. Rheum. Dis. 53, 515–520 (1994).
Hallett, M. B. & Lloyds, D. Neutrophil priming: the cellular signals that say 'amber' but not 'green'. Immunol. Today 16, 264–268 (1995).
Fossati, G., Moots, R. J., Bucknall, R. C. & Edwards, S. W. Differential role of neutrophil Fcγ receptor IIIB (CD16) in phagocytosis, bacterial killing, and responses to immune complexes. Arthritis Rheum. 46, 1351–1361 (2002).
Lefrancais, E. et al. IL-33 is processed into mature bioactive forms by neutrophil elastase and cathepsin G. Proc. Natl Acad. Sci. USA 109, 1673–1678 (2012).
Van den Steen, P. E. et al. Cleavage of denatured natural collagen type II by neutrophil gelatinase B reveals enzyme specificity, post-translational modifications in the substrate, and the formation of remnant epitopes in rheumatoid arthritis. FASEB J. 16, 379–389 (2002).
Katano, M. et al. Implication of granulocyte-macrophage colony-stimulating factor induced neutrophil gelatinase-associated lipocalin in pathogenesis of rheumatoid arthritis revealed by proteome analysis. Arthritis Res. Ther. 11, R3 (2009).
Elsaid, K. A., Jay, G. D. & Chichester, C. O. Detection of collagen type II and proteoglycans in the synovial fluids of patients diagnosed with non-infectious knee joint synovitis indicates early damage to the articular cartilage matrix. Osteoarthritis Cartilage 11, 673–680 (2003).
Baici, A., Salgam, P., Cohen, G., Fehr, K. & Boni, A. Action of collagenase and elastase from human polymorphonuclear leukocytes on human articular cartilage. Rheumatol. Int. 2, 11–16 (1982).
Oseas, R., Yang, H. H., Baehner, R. L. & Boxer, L. A. Lactoferrin: a promoter of polymorphonuclear leukocyte adhesiveness. Blood 57, 939–945 (1981).
Sopata, I. et al. Neutrophil gelatinase levels in plasma and synovial fluid of patients with rheumatic diseases. Rheumatol. Int. 15, 9–14 (1995).
Pham, C. T. Neutrophil serine proteases: specific regulators of inflammation. Nat. Rev. Immunol. 6, 541–550 (2006).
Wang, C. H. et al. Expression of CD147 (EMMPRIN) on neutrophils in rheumatoid arthritis enhances chemotaxis, matrix metalloproteinase production and invasiveness of synoviocytes. J. Cell. Mol. Med. 15, 850–860 (2011).
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Khandpur, R. et al. NETs are a source of citrullinated autoantigens and stimulate inflammatory responses in rheumatoid arthritis. Sci. Transl. Med. 5, 178ra40 (2013).
Baillet, A. et al. Synovial fluid proteomic fingerprint: S100A8, S100A9 and S100A12 proteins discriminate rheumatoid arthritis from other inflammatory joint diseases. Rheumatology 49, 671–682 (2010).
Simard, J. C., Girard, D. & Tessier, P. A. Induction of neutrophil degranulation by S100A9 via a MAPK-dependent mechanism. J. Leukoc. Biol. 87, 905–914 (2010).
Laurindo, I. M., Mello, S. B. & Cossermelli, W. Influence of low doses of methotrexate on superoxide anion production by polymorphonuclear leukocytes from patients with rheumatoid arthritis. J. Rheumatol. 22, 633–638 (1995).
Kraan, M. C. et al. Inhibition of neutrophil migration soon after initiation of treatment with leflunomide or methotrexate in patients with rheumatoid arthritis: findings in a prospective, randomized, double-blind clinical trial in fifteen patients. Arthritis Rheum. 43, 1488–1495 (2000).
Sperling, R. I. et al. Acute and chronic suppression of leukotriene B4 synthesis ex vivo in neutrophils from patients with rheumatoid arthritis beginning treatment with methotrexate. Arthritis Rheum. 35, 376–384 (1992).
Elferink, J. G., Daha, M. R. & de Koster, B. M. A cyclic GMP- and G-kinase-dependent effect of azathioprine on migration by human neutrophils. Cell. Mol. Life Sci. 53, 593–599 (1997).
Akahoshi, T. et al. Rapid induction of neutrophil apoptosis by sulfasalazine: implications of reactive oxygen species in the apoptotic process. J. Leukoc. Biol. 62, 817–826 (1997).
Wandall, J. H. Effects of sulphasalazine and its metabolites on neutrophil chemotaxis, superoxide production, degranulation and translocation of cytochrome b-245. Aliment. Pharmacol. Ther. 5, 609–619 (1991).
Cross, A., Moots, R. J. & Edwards, S. W. The dual effects of TNFα on neutrophil apoptosis are mediated via differential effects on expression of Mcl-1 and Bfl-1. Blood 111, 878–884 (2008).
Ginis, I. & Tauber, A. I. Activation mechanisms of adherent human neutrophils. Blood 76, 1233–1239 (1990).
Wipke, B. T. & Allen, P. M. Essential role of neutrophils in the initiation and progression of a murine model of rheumatoid arthritis. J. Immunol. 167, 1601–1608 (2001).
Tanaka, D., Kagari, T., Doi, H. & Shimozato, T. Essential role of neutrophils in anti-type II collagen antibody and lipopolysaccharide-induced arthritis. Immunology 119, 195–202 (2006).
Coelho, F. M. et al. The chemokine receptors CXCR1/CXCR2 modulate antigen-induced arthritis by regulating adhesion of neutrophils to the synovial microvasculature. Arthritis Rheum. 58, 2329–2337 (2008).
Grespan, R. et al. CXCR2-specific chemokines mediate leukotriene B4-dependent recruitment of neutrophils to inflamed joints in mice with antigen-induced arthritis. Arthritis Rheum. 58, 2030–2040 (2008).
Barsante, M. M. et al. Blockade of the chemokine receptor CXCR2 ameliorates adjuvant-induced arthritis in rats. Br. J. Pharmacol. 153, 992–1002 (2008).
Min., S. H. et al. Pharmacological targeting reveals distinct roles for CXCR2/CXCR1 and CCR2 in a mouse model of arthritis. Biochem. Biophys. Res. Commun. 391, 1080–1086 (2009).
Cunha, T. M. et al. Treatment with DF 2162, a non-competitive allosteric inhibitor of CXCR1/2, diminishes neutrophil influx and inflammatory hypernociception in mice. Br. J. Pharmacol. 154, 460–470 (2008).
Grant, E. P. et al. Essential role for the C5a receptor in regulating the effector phase of synovial infiltration and joint destruction in experimental arthritis. J. Exp. Med. 196, 1461–1471 (2002).
Tsuboi, N. et al. Regulation of human neutrophil Fcγ receptor IIa by C5a receptor promotes inflammatory arthritis in mice. Arthritis Rheum. 63, 467–478 (2011).
Sadik, C. D., Kim, N. D., Iwakura, Y. & Luster, A. D. Neutrophils orchestrate their own recruitment in murine arthritis through C5aR and FcγR signaling. Proc. Natl Acad. Sci. USA 109, E3177–E3185 (2012).
Kim, N. D., Chou, R. C., Seung, E., Tager, A. M. & Luster, A. D. A unique requirement for the leukotriene B4 receptor BLT1 for neutrophil recruitment in inflammatory arthritis. J. Exp. Med. 203, 829–835 (2006).
Grevers, L. C. et al. Different amplifying mechanisms of interleukin-17 and interferon-γ in Fcγ receptor-mediated cartilage destruction in murine immune complex-mediated arthritis. Arthritis Rheum. 60, 396–407 (2009).
Damsker, J. M. et al. Targeting the chemotactic function of CD147 reduces collagen-induced arthritis. Immunology 126, 55–62 (2009).
Sarraj, B., Ludanyi, K., Glant, T. T., Finnegan, A. & Mikecz, K. Expression of CD44 and L-selectin in the innate immune system is required for severe joint inflammation in the proteoglycan-induced murine model of rheumatoid arthritis. J. Immunol. 177, 1932–1940 (2006).
Eyles, J. L. et al. A key role for G-CSF-induced neutrophil production and trafficking during inflammatory arthritis. Blood 112, 5193–5201 (2008).
Williams, A. S. et al. Interferon-γ protects against the development of structural damage in experimental arthritis by regulating polymorphonuclear neutrophil influx into diseased joints. Arthritis Rheum. 56, 2244–2254 (2007).
Kelchtermans, H. et al. Effector mechanisms of interleukin-17 in collagen-induced arthritis in the absence of interferon-γ and counteraction by interferon-γ. Arthritis Res. Ther. 11, R122 (2009).
Davey, M. S. et al. Failure to detect production of IL-10 by activated human neutrophils. Nat. Immunol. 12, 1017–1020 (2011).
Mestas, J. & Hughes, C. C. Of mice and not men: differences between mouse and human immunology. J. Immunol. 172, 2731–2738 (2004).
Fuchs, T. A. et al. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 176, 231–241 (2007).
Branzk, N. & Papayannopoulos, V. Molecular mechanisms regulating NETosis in infection and disease. Semin. Immunopathol. 35, 513–530 (2013).
Cooper, P. R., Palmer, L. J. & Chapple, I. L. Neutrophil extracellular traps as a new paradigm in innate immunity: friend or foe? Periodontol. 2000 63, 165–197 (2013).
Yousefi, S., Mihalache, C., Kozlowski, E., Schmid, I. & Simon, H. U. Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps. Cell Death Differ. 16, 1438–1444 (2009).
Wang, Y. et al. Histone hypercitrullination mediates chromatin decondensation and neutrophil extracellular trap formation. J. Cell Biol. 184, 205–213 (2009).
Knight, J. S., Carmona-Rivera, C. & Kaplan, M. J. Proteins derived from neutrophil extracellular traps may serve as self-antigens and mediate organ damage in autoimmune diseases. Front. Immunol. 3, 380 (2012).
Garcia-Romo, G. S. et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci. Transl. Med. 3, 73ra20 (2011).
Lande, R. et al. Neutrophils activate plasmacytoid dendritic cells by releasing self–DNA–peptide complexes in systemic lupus erythematosus. Sci. Transl. Med. 3, 73ra19 (2011).
Villanueva, E. et al. Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J. Immunol. 187, 538–552 (2011).
Kaplan, M. J. Neutrophils in the pathogenesis and manifestations of SLE. Nat. Rev. Rheumatol. 7, 691–699 (2011).
Midgley, A., McLaren, Z., Moots, R. J., Edwards, S. W. & Beresford, M. W. The role of neutrophil apoptosis in juvenile-onset systemic lupus erythematosus. Arthritis Rheum. 60, 2390–2401 (2009).
Hacbarth, E. & Kajdacsy-Balla, A. Low density neutrophils in patients with systemic lupus erythematosus, rheumatoid arthritis, and acute rheumatic fever. Arthritis Rheum. 29, 1334–1342 (1986).
Denny, M. F. et al. A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs. J. Immunol. 184, 3284–3297 (2010).
Theilgaard-Monch, K. et al. The transcriptional program of terminal granulocytic differentiation. Blood 105, 1785–1796 (2005).
Hakkim, A. et al. Impairment of neutrophil extracellular trap degradation is associated with lupus nephritis. Proc. Natl Acad. Sci. USA 107, 9813–9818 (2010).
Romero, V. et al. Immune-mediated pore-forming pathways induce cellular hypercitrullination and generate citrullinated autoantigens in rheumatoid arthritis. Sci. Transl. Med. 5, 209ra150 (2013).
Scally, S. W. et al. A molecular basis for the association of the HLA-DRB1 locus, citrullination, and rheumatoid arthritis. J. Exp. Med. 210, 2569–2582 (2013).
Pratesi, F. et al. Antibodies from patients with rheumatoid arthritis target citrullinated histone 4 contained in neutrophils extracellular traps. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2012-202765.
Acknowledgements
The authors thank Dr Will Boswell for provision of the MRI scan in Figure 1. H.L.W.'s work is supported by grant number 19437 from Arthritis Research UK.
Author information
Authors and Affiliations
Contributions
H.L.W. and S.W.E. researched the data for article and wrote the manuscript. All authors (H.L.W., R.J.M. and S.W.E.) contributed substantially to discussions of the article content, and review or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Wright, H., Moots, R. & Edwards, S. The multifactorial role of neutrophils in rheumatoid arthritis. Nat Rev Rheumatol 10, 593–601 (2014). https://doi.org/10.1038/nrrheum.2014.80
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.80
This article is cited by
-
Study of serum calprotectin level in rheumatoid arthritis: unexpected low level and possible explanations
Egyptian Rheumatology and Rehabilitation (2024)
-
Peptide-anchored neutrophil membrane-coated biomimetic nanodrug for targeted treatment of rheumatoid arthritis
Journal of Nanobiotechnology (2023)
-
Glutathione peroxidase 3 is a novel clinical diagnostic biomarker and potential therapeutic target for neutrophils in rheumatoid arthritis
Arthritis Research & Therapy (2023)
-
Radiographic airway abnormalities in untreated early rheumatoid arthritis are associated with peripheral neutrophil activation
Arthritis Research & Therapy (2023)
-
Targeting the activity of T cells by membrane surface redox regulation for cancer theranostics
Nature Nanotechnology (2023)