Abstract
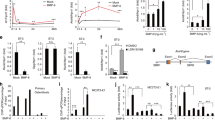
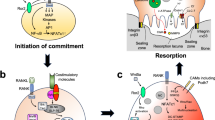
The signaling molecule Wnt regulates bone homeostasis through β-catenin–dependent canonical and β-catenin–independent noncanonical pathways. Impairment of canonical Wnt signaling causes bone loss in arthritis and osteoporosis; however, it is unclear how noncanonical Wnt signaling regulates bone resorption. Wnt5a activates noncanonical Wnt signaling through receptor tyrosine kinase-like orphan receptor (Ror) proteins. We showed that Wnt5a-Ror2 signaling between osteoblast-lineage cells and osteoclast precursors enhanced osteoclastogenesis. Osteoblast-lineage cells expressed Wnt5a, whereas osteoclast precursors expressed Ror2. Mice deficient in either Wnt5a or Ror2, and those with either osteoclast precursor-specific Ror2 deficiency or osteoblast-lineage cell-specific Wnt5a deficiency showed impaired osteoclastogenesis. Wnt5a-Ror2 signals enhanced receptor activator of nuclear factor-κB (RANK) expression in osteoclast precursors by activating JNK and recruiting c-Jun on the promoter of the gene encoding RANK, thereby enhancing RANK ligand (RANKL)-induced osteoclastogenesis. A soluble form of Ror2 acted as a decoy receptor of Wnt5a and abrogated bone destruction in mouse arthritis models. Our results suggest that the Wnt5a-Ror2 pathway is crucial for osteoclastogenesis in physiological and pathological environments and represents a therapeutic target for bone diseases, including arthritis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Teitelbaum, S.L. & Ross, F.P. Genetic regulation of osteoclast development and function. Nat. Rev. Genet. 4, 638–649 (2003).
Takahashi, N. et al. Osteoblastic cells are involved in osteoclast formation. Endocrinology 123, 2600–2602 (1988).
Yasuda, H. et al. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. USA 95, 3597–3602 (1998).
Lacey, D.L. et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 93, 165–176 (1998).
Simonet, W.S. et al. Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell 89, 309–319 (1997).
Boyle, W.J., Simonet, W.S. & Lacey, D.L. Osteoclast differentiation and activation. Nature 423, 337–342 (2003).
Suda, T. et al. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 20, 345–357 (1999).
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Calvi, L.M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Arai, F. et al. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell 118, 149–161 (2004).
Wu, J.Y. et al. Osteoblastic regulation of B lymphopoiesis is mediated by Gsa-dependent signaling pathways. Proc. Natl. Acad. Sci. USA 105, 16976–16981 (2008).
Mizoguchi, T. et al. Identification of cell cycle-arrested quiescent osteoclast precursors in vivo. J. Cell Biol. 184, 541–554 (2009).
Bienz, M. & Clevers, H. Linking colorectal cancer to Wnt signaling. Cell 103, 311–320 (2000).
Miller, J.R. The Wnts. Genome Biol. 3, 3001.1–3001.15 (2001).
Logan, C.Y. & Nusse, R. The Wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol. 20, 781–810 (2004).
Staal, F.J., Luis, T.C. & Tiemessen, M.M. WNT signaling in the immune system: WNT is spreading its wings. Nat. Rev. Immunol. 8, 581–593 (2008).
Day, T.F. et al. Wnt/β-catenin signaling in mesenchymal progenitors controls osteoblast and chondrocyte differentiation during vertebrate skeletogenesis. Dev. Cell 8, 739–750 (2005).
Gong, Y. et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 107, 513–523 (2001).
Kato, M. et al. Cbfa1-independent decrease in osteoblast proliferation, osteopenia, and persistent embryonic eye vascularization in mice deficient in Lrp5, a Wnt coreceptor. J. Cell Biol. 157, 303–314 (2002).
Baron, R. & Rawadi, G. Targeting the Wnt/β-catenin pathway to regulate bone formation in the adult skeleton. Endocrinology 148, 2635–2643 (2007).
Yadav, V.K. et al. Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell 135, 825–837 (2008).
Cui, Y. et al. Lrp5 functions in bone to regulate bone mass. Nat. Med. 17, 684–691 (2011).
Glass, D.A. 2nd et al. Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Dev. Cell 8, 751–764 (2005).
Ishitani, T. et al. The TAK1-NLK mitogen-activated protein kinase cascade functions in the Wnt-5a/Ca2+ pathway to antagonize Wnt/β-catenin signaling. Mol. Cell. Biol. 23, 131–139 (2003).
Liang, H. et al. Wnt5a inhibits B cell proliferation and functions as a tumor suppressor in hematopoietic tissue. Cancer Cell 4, 349–360 (2003).
Oishi, I. et al. The receptor tyrosine kinase Ror2 is involved in non-canonical Wnt5a/JNK signalling pathway. Genes Cells 8, 645–654 (2003).
Billiard, J. et al. The orphan receptor tyrosine kinase Ror2 modulates canonical Wnt signaling in osteoblastic cells. Mol. Endocrinol. 19, 90–101 (2005).
Liu, Y. et al. The orphan receptor tyrosine kinase Ror2 promotes osteoblast differentiation and enhances ex vivo bone formation. Mol. Endocrinol. 21, 376–387 (2007).
Goldring, S.R. & Gravallese, E.M. Pathogenesis of bone erosions in rheumatoid arthritis. Curr. Opin. Rheumatol. 12, 195–199 (2000).
Li, X. et al. Dkk2 has a role in terminal osteoblast differentiation and mineralized matrix formation. Nat. Genet. 37, 945–952 (2005).
Zhou, H., Mak, W., Zheng, Y., Dunstan, C.R. & Seibel, M.J. Osteoblasts directly control lineage commitment of mesenchymal progenitor cells through Wnt signaling. J. Biol. Chem. 283, 1936–1945 (2008).
Yamaguchi, T.P., Bradley, A., McMahon, A.P. & Jones, S.A. Wnt5a pathway underlies outgrowth of multiple structures in the vertebrate embryo. Development 126, 1211–1223 (1999).
DeChiara, T.M. et al. Ror2, encoding a receptor-like tyrosine kinase, is required for cartilage and growth plate development. Nat. Genet. 24, 271–274 (2000).
Takeuchi, S. et al. Mouse Ror2 receptor tyrosine kinase is required for the heart development and limb formation. Genes Cells 5, 71–78 (2000).
Yamane, T. et al. Wnt signaling regulates hemopoiesis through stromal cells. J. Immunol. 167, 765–772 (2001).
Takada, I. et al. A histone lysine methyltransferase activated by non-canonical Wnt signaling suppresses PPAR-γ transactivation. Nat. Cell Biol. 9, 1273–1285 (2007).
Nakamura, T. et al. Estrogen prevents bone loss via estrogen receptor α and induction of Fas ligand in osteoclasts. Cell 130, 811–823 (2007).
Rodda, S.J. & McMahon, A.P. Distinct roles for Hedgehog and canonical Wnt signaling in specification, differentiation and maintenance of osteoblast progenitors. Development 133, 3231–3244 (2006).
Kwon, O.H., Lee, C.-K., Lee, Y.I., Paik, S.-G. & Lee, H.-J. The hematopoietic transcription factor PU.1 regulates RANK gene expression in myeloid progenitors. Biochem. Biophys. Res. Commun. 335, 437–446 (2005).
Kardassis, D., Papakosta, P., Pardali, K. & Moustakas, A. c-Jun transactivates the promoter of the human p21(WAF1/Cip1) gene by acting as a superactivator of the ubiquitous transcription factor Sp1. J. Biol. Chem. 274, 29572–29581 (1999).
Blaine, S.A., Wick, M., Dessev, C. & Nemenoff, R.A. Induction of cPLA2 in lung epithelial cells and non-small cell lung cancer is mediated by Sp1 and c-Jun. J. Biol. Chem. 276, 42737–42743 (2001).
Sen, M. et al. Expression and function of wingless and frizzled homologs in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 97, 2791–2796 (2000).
Diarra, D. et al. Dickkopf-1 is a master regulator of joint remodeling. Nat. Med. 13, 156–163 (2007).
Kim, N., Takami, M., Rho, J., Josien, R. & Choi, Y. A novel member of the leukocyte receptor complex regulates osteoclast differentiation. J. Exp. Med. 195, 201–209 (2002).
Koga, T. et al. Costimulatory signals mediated by the ITAM motif cooperate with RANKL for bone homeostasis. Nature 428, 758–763 (2004).
Nakashima, T. et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 17, 1231–1234 (2011).
Xiong, J. et al. Matrix-embedded cells control osteoclast formation. Nat. Med. 17, 1235–1241 (2011).
Arai, F. et al. Commitment and differentiation of osteoclast precursor cells by the sequential expression of c-Fms and receptor activator of nuclear factor κB (RANK) receptors. J. Exp. Med. 190, 1741–1754 (1999).
Schett, G., Zwerina, J. & David, J.P. The role of Wnt proteins in arthritis. Nat. Clin. Pract. Rheumatol. 4, 473–480 (2008).
Topol, L. et al. Wnt-5a inhibits the canonical Wnt pathway by promoting GSK-3-independent β-catenin degradation. J. Cell Biol. 162, 899–908 (2003).
Sato, A., Yamamoto, H., Sakane, H., Koyama, H. & Kikuchi, A. Wnt5a regulates distinct signalling pathways by binding to Frizzled2. EMBO J. 29, 41–54 (2010).
Pettit, A.R., Walsh, N.C., Manning, C., Goldring, S.R. & Gravallese, E.M. RANKL protein is expressed at the pannus-bone interface at sites of articular bone erosion in rheumatoid arthritis. Rheumatology 45, 1068–1076 (2006).
Kubota, A., Hasegawa, K., Suguro, T. & Koshihara, Y. Tumor necrosis factor-α promotes the expression of osteoprotegerin in rheumatoid synovial fibroblasts. J. Rheumatol. 31, 426–435 (2004).
Afzal, A.R. et al. Recessive Robinow syndrome, allelic to dominant brachydactyly type B, is caused by mutation of ROR2. Nat. Genet. 25, 419–422 (2000).
van Bokhoven, H. et al. Mutation of the gene encoding the ROR2 tyrosine kinase causes autosomal recessive Robinow syndrome. Nat. Genet. 25, 423–426 (2000).
Nomi, M. et al. Loss of mRor1 enhances the heart and skeletal abnormalities in mRor2-deficient mice: redundant and pleiotropic functions of mRor1 and mRor2 receptor tyrosine kinases. Mol. Cell. Biol. 21, 8329–8335 (2001).
Binder, N.B. et al. Estrogen-dependent and C-C chemokine receptor-2–dependent pathways determine osteoclast behavior in osteoporosis. Nat. Med. 15, 417–424 (2009).
Morita, S., Kojima, T. & Kitamura, T. Plat-E: an efficient and stable system for transient packaging of retroviruses. Gene Ther. 7, 1063–1066 (2000).
Trentham, D.E., Townes, A.S. & Kang, A.H. Autoimmunity to type II collagen an experimental model of arthritis. J. Exp. Med. 146, 857–868 (1977).
Pettit, A.R. et al. TRANCE/RANKL knockout mice are protected from bone erosion in a serum transfer model of arthritis. Am. J. Pathol. 159, 1689–1699 (2001).
Acknowledgements
We thank T. Kitamura (Tokyo University) for pMX-IRES-GFP vectors and Plat-E cells; M. Sakoda, S. Kinugawa, Y. Nakamichi and T. Yamashita for technical assistance; A. Yamaguchi, H. Takayanagi and T. Nakashima for thoughtful discussion; and T. Nishizawa, Y. Ueno and M. Saito for discussions about mice models. This work was supported by a Grant-in-Aid for Scientific Research (18390495, 22390351 (N.T.); 18390557, 21390551 (Y. Kobayashi); 19390476, 21390498 (N.U.)) from the Ministry of Education, Culture, Sports, Science and Technology of Japan and by a grant from the Naito Foundation (Y. Kobayashi).
Author information
Authors and Affiliations
Contributions
K.M. and Y. Kobayashi contributed equally to the manuscript, conducting most of the experiments and cooperating in the preparation of the manuscript. N.U., S.U., I.T., S. Kato, K.M. and T.J.M. supported the in vivo experiments, conducting the data analysis, and contributed to the manuscript preparation. T.M., A.I. and Y. Kikuchi carried out the immunohistochemistry and purified recombinant proteins. S. Kani, M.N. and Y.M. generated Ror2-deficient, Ror2-floxed mice and Ror2-specific antibodies and contributed to the data analysis. Y. Kobayashi generated Tnfrsf11aCre/+ and Wnt5a-floxed mice. Y. Kobayashi and N.T. designed and supervised the project and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Methods (PDF 959 kb)
Rights and permissions
About this article
Cite this article
Maeda, K., Kobayashi, Y., Udagawa, N. et al. Wnt5a-Ror2 signaling between osteoblast-lineage cells and osteoclast precursors enhances osteoclastogenesis. Nat Med 18, 405–412 (2012). https://doi.org/10.1038/nm.2653
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2653
This article is cited by
-
WNT5A regulates the proliferation, apoptosis and stemness of human stem Leydig cells via the β-catenin signaling pathway
Cellular and Molecular Life Sciences (2024)
-
Role of Wnt signaling and sclerostin in bone and as therapeutic targets in skeletal disorders
Osteoporosis International (2023)
-
Quantitative analyses of matrices, osteoblasts, and osteoclasts during bone remodeling using an in vitro system
Journal of Bone and Mineral Metabolism (2023)
-
Osteoclast biology in the single-cell era
Inflammation and Regeneration (2022)
-
The effects and mechanism of paeoniflorin in promoting osteogenic differentiation of MC3T3-E1
Journal of Orthopaedic Surgery and Research (2022)