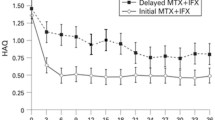
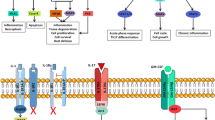
Summary
Tumour necrosis factor-α (TNFα) is a pleiotropic cytokine which is overproduced in rheumatoid joints primarily by macrophages. This cytokine has a potentialpathogenic role in the establishment of rheumatoid synovitis, in the formationof pannus tissue and in the process of joint destruction, as it increases synoviocyte proliferation and triggers a cascade of secondary mediators involved in the recruitment of inflammatory cells, in neo-angiogenesis and in the process of joint destruction. These findings made TNFα a potential target for anticytokine therapy.Experimental studies have shown that TNFα blockade by monoclonal antibodies or by soluble TNF receptor reduced the extent and severity of arthritis bothin collagen-induced arthritis in mice and in transgenic mice overexpressingTNFα, which develop a rheumatoid-like destructive arthritis. Clinical studiesbased on the use of anti-TNFα antibodies or soluble receptors have suggested a potential beneficial effect of TNFα-blocking therapy in inducing amelioration of inflammatory parameters in patients with long-standing active disease. In these patients anti-TNFα therapy induces a rapid improvement in multiple clinical assessment of disease activity, including morning stiffness, pain score, Ritchie articular index and swollen joint count. The clinical benefits are associated withan improvement in some serological parameters, such as C-reactive protein and serum amyloid-A, erythrocyte sedimentation rate, blood cytokine levels, haemoglobin, white cells and platelet counts, rheumatoid factor titre and histological features of the synovium. However, it remains to be determined whether anti-TNFα therapy may be useful in the long term management of rheumatoid patients and in the achievement of better outcomes of disease. Because TNFα production also serves a specific function in host defence against infections and tumours, the adverse effects of long term anti-TNFα therapy must be carefully evaluated. In addition, targeting a single mediator may be not sufficient to block the complex inflammatory response in rheumatoid arthritis. For these reasons therapeutic strategies aimed at concomitantly interfering with multiple pathogenic pathways are currently under investigation.
Similar content being viewed by others
References
Silman AJ, Hockberg MC. Epidemiology of rheumatic diseases. Oxford: Oxford University Press, 1993
Erhardt CC, Mumford PA, Venables PJ, et al. Factors predicting a poor life prognosis in rheumatoid arthritis: an eight year prospective study. Ann Rheum Dis 1989; 48: 7–13
Feldmann M, Brennan FM, Maini RN. Role of cytokines in rheumatoid arthritis. Ann Rev Immunol 1996; 14: 397–440
Feldmann M, Elliott MJ, Woody JN, et al. Anti-tumor necrosis factor-α therapy of rheumatoid arthritis. Adv Immunol 1997; 64: 283–350
Tetta C, Camussi G, Modena V, et al. Tumor necrosis factor in serum and synovial fluid of patients with active and severe rheumatoid arthritis. Ann Rheum Dis 1990; 49: 665–7
Brennan FN, Gibbons DL, Mitchell T, et al. Enhanced expression of tumor necrosis factor receptor mRNA and protein in mononuclear cells isolated from rheumatoid arthritis synovial joints. Eur J Immunol 1992; 22: 1907–12
Husby G, Williams RC. Synovial localization of tumor necrosis factor in patients with rheumatoid arthritis. J Autoimmun 1988; 1: 363–71
Lupia E, Montrucchio G, Battaglia E, et al. Role of tumor necrosis factor-a and platelet-activating factor in neoangiogenesis induced by synovial fluids of patients with rheumatoid arthritis. Eur J Immunol 1996; 26: 1690–4
Dayer JM, Beutler B, Cerami A. Cachectin/tumor necrosis factor stimulates collagenase and prostaglandin E production by human synovial cells and dermal fibroblasts. J Exp Med 1985; 162: 2163–8
Deleuran BMW, Chu CQ, Field M, et al. Localization of tumor necrosis factor receptors in the synovial tissue and cartilage-pannus junction in patients with rheumatoid arthritis: implications for local actions of tumor necrosis factor α. Arthritis Rheum 1992; 35: 1170–8
Keffer J, Probert L, Cazlaris H, et al. Transgenic mice expressing human tumor necrosis factor: a predictive genetic model of arthritis. EMBO J 1991; 10: 4025–31
Piquet PF, Grau GE, Vesin C, et al. Evolution of collagen arthritis in mice is arrested by treatment with anti-tumor necrosis factor (TNF) antibody or a recombinant soluble TNF receptor. Immunology 1992; 77: 510–4
Wooley PH, Dutcher J, Widmer MB, et al. Influence of a recombinant human soluble tumor necrosis factor receptor Fc fusion protein on type II collagen-induced arthritis in mice. J Immunol 1993; 151: 6602–7
Firestein GS, Zvaifler NJ. Anticytokine therapy in rheumatoid arthritis. N Engl J Med 1997; 337: 195–7
Knight DM, Trinh H, Le J, et al. Construction and initial characterization of a mouse-human chimeric anti-TNF antibody. Mol Immunol 1993; 30: 1443–53
Elliott MJ, Maini RN, Feldmann M, et al. Randomized double-blind comparison of chimeric monoclonal antibody to tumor necrosis factor-α (cA2) versus placebo in rheumatoid arthritis. Lancet 1994; 344: 1105–10
Paulus HE, Egger MJ, Ward JR, et al. Analysis of improvement in individual rheumatoid arthritis patients treated with disease-modifying antirheumatic drugs, based on the findings in patients treated with placebo. Arthritis Rheum 1990; 33: 477–84
Elliott MJ, Maini RN, Feldmann M, et al. Repeated therapy with monoclonal antibody to tumor necrosis factor-α (cA2) in patients with rheumatoid arthritis. Lancet 1994; 344: 1125–8
Elliott MJ, Maini RN, Feldmann M, et al. Treatment of rheumatoid arthritis with chimeric monoclonal antibodies to tumor necrosis factor-α. Arthritis Rheum 1993; 36: 1681–90
Lorenz H-M, Antoni C, Valerius T, et al. In vivo blockade of TNF-α by intravenous infusion of a chimeric monoclonal TNF-α antibody in patients with rheumatoid arthritis: short term cellular and molecular effects. J Immunol 1996; 156: 1646–53
Tak PP, Taylor PC, Breedveld FC, et al. Decrease in cellularity and expression of adhesion molecules by anti-tumor necrosis factor-α monoclonal antibody treatment in patients with rheumatoid arthritis. Arthritis Rheum 1996; 39: 1077–81
Paleolog EM, Hunt M, Elliott MJ, et al. Deactivation of vascular endothelium by monoclonal anti-tumor necrosis factor-α antibody in rheumatoid arthritis. Arthritis Rheum 1996; 39: 1082–91
Rankin ECC, Choy EHS, Kassimos D, et al. The therapeutic effects of an engineered human anti-tumor necrosis factor alpha antibody (CDP571) in rheumatoid arthritis. Br J Rheumatol 1995; 34: 334–42
Moreland LW, Margolies G, Heck Jr LW, et al. Recombinant soluble tumor necrosis factor receptor (p80) fusion protein: toxicity and dose finding trial in refractory rheumatoid arthritis. J Rheumatol 1996; 23: 1849–55
Moreland LW, Baumgartner SW, Schiff MH, et al. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)-Fc fusion protein. N Engl J Med 1997; 337: 141–7
Epstein WV. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor-Fc fusion protein. N Engl J Med 1997; 337: 337–8
Williams RO, Mason LJ, Feldmann M, et al. Synergy between anti-CD4 and anti-tumor necrosis factor in the amelioration of established collagen-induced arthritis. Proc Natl Acad Sci USA 1994; 91: 2762–6
Linsley PS, Wallace PM, Johnson J, et al. Immunosuppression in vivo by a soluble form of the CTLA-4 T cell activation molecule. Science 1992; 257: 792–5
Biancone L, Andres G, Ahn A, et al. Inhibition of the CD40-CD40 ligand pathway prevents murine membranous glomer-ulonephritis. Kidney Int 1995; 48: 458–68
Mannik M, Wener M. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor-Fc fusion protein. N Engl J Med 1997; 337: 338
Gay S, Gay RE, Koopman WJ. Molecular and cellular mechanisms of joint destruction in rheumatoid arthritis: two cellular mechanisms explain joint destruction. Ann Rheum Dis 1993; 52: S39–47
Muller-Ladner U, Kriegsmann J, Franklin BN, et al. Synovial fibroblasts of patients with rheumatoid arthritis attach to and invade normal human cartilage when engrafted into SCID mice. Am J Pathol 1996; 149: 1607–15
Russell D, Tucker K, Kohno H, et al. Inhibition of tumor necrosis factor is effective alone or in combination with inhibition of interleukin-1 in reducing joint swelling in rodent bacterial cell wall-induced arthritis. Arthritis Rheum 1994; 37 Suppl. 9: 279
Joyce DA, Gibbons DP, Green P, et al. Two inhibitors of proinflammatory cytokine release, interleukin-10 and interleukin-4, have contrasting effects on release of soluble p75 tumor necrosis factor receptor by cultured monocytes. Eur J Immunol 1994; 24: 2699–705
Joosten LA, Lubberts E, Durez P, et al. Role of interleukin-4 and interleukin-10 in murine collagen-induced arthritis: protective effect of interleukin-4 and interleukin-10 treatment on cartilage destruction. Arthritis Rheum 1997; 40: 249–60
Le CH, Nicolson G, Morales A, et al. Suppression of collagen-induced arthritis through adenovirus-mediated transfer of a modified tumor-necrosis factor a receptor gene. Arthritis Rheum 1997; 40: 1662–9
Montrucchio G, Lupia E, De Martino A, et al. Nitric oxide mediates angiogenesis induced in vivo by platelet-activating factor and tumor necrosis factor but not by basic fibroblast growth factor. Am J Pathol 1997; 151: 557–63
Huizinga TW, Dijkmans BA, van der Velde EA, et al. An open study of pentoxifylline and thalidomide as adjuvant therapy in the treatment of rheumatoid arthritis. Ann Rheum Dis 1996; 55: 833–6
Maksymowych WP, Avina-Zubieta A, Luong MH, et al. An open study of pentoxifylline in the treatment of severe refractory rheumatoid arthritis. J Rheumatol 1995; 22: 625–9
van der Borne BE, Dijkmans BA, de Rooij HH, et al. Chloroquine and hydroxychloroquine equally affect tumor necrosis factor-alfa, interleukine 6, and interferon-gamma production by peripheral blood mononuclear cells. J Rheumatol 1997; 24: 55–60
Moss ML, Jin SL, Milla ME, et al. Cloning a disintegrin metallo-proteinase that processes precursor tumor-necrosis factor-alpha. Nature 1997; 385: 733–6
Black RA, Rauch CT, Kozlosky CJ, et al. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 1997; 385: 729–33
Williams LM, Gibbons DL, Geargin A, et al. Paradoxical effects of a synthetic metalloproteinase inhibitor that blocks both p55 and p75 TNF receptor shedding and TNF alpha processing in RA synovial membrane cell cultures. J Clin Invest 1996; 97: 2833–41
Sander O, den Broeder A, van der Hoogen, et al. 30 months of treatment with a TNFα receptor-fusion protein (TNFR55-IGG1, RO 45-2081) in patients with severe refractory RA [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s81
Sander O, Bock E, Rau R. Safety and efficacy of TNFa receptor-fusion protein (TNFR55-IGG1, RO 45-2081) in patients with RA and liver cirrhosis [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s81
Weisman MH, Markenson J, Wiik A, et al. TNF neutralization does not cause autoantibody changes: the experience with 521z patients enrolled into IV RO 45-2081 trials [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s81
Morgan AW, Hale G, Rebello P, et al. Combination therapy with a TNF antagonist and a CD4 monoclonal antibody in rheumatoid arthritis. A pilot study [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s81
Minghetti PP, Drlica K, Zhang HG, et al. TNFα ribozymes and antisense molecules as potential therapeutic agents for treatment of rheumatoid arthritis [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s81
Maini RN, Breedveld FC, Kalden JR, et al. Low dose methotrexate suppresses anti-globulin responses and potentiates efficacy of a chimeric monoclonal anti-TNFα antibody (cA2) given repeatedly in rheumatoid arthritis [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: sl26
Weinblatt M, Schiff MH, Baumgartner SW, et al. Long term and phase III treatment of dmard failing rheumatoid arthritis patients with TNF receptor P75 FC fusion protein (TNFR; FC; ENBREL) [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: sl26
Sander O, Rau R. Long term observation of 80 patients treated with TNFα receptor-fusion protein (TNFR55-IGG1, RO 45-2081) [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s224
Evans J, Yocum DE, Maricic M, et al. Results of an open label, dose-ranging, safety and pharmacokinetic trial of the humanized tumor necrosis factor antibody (HTA) bay 103356, in patients with active rheumatoid arthritis [abstract]. Arthritis Rheum 1997; 40 Suppl. 9: s224
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Camussi, G., Lupia, E. The Future Role of Anti-Tumour Necrosis Factor (TNF) Products in the Treatment of Rheumatoid Arthritis. Drugs 55, 613–620 (1998). https://doi.org/10.2165/00003495-199855050-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199855050-00001