Abstract
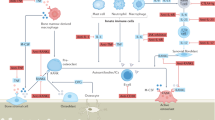
The introduction of targeted therapies has dramatically changed the prognosis of patients with chronic joint diseases such as rheumatoid arthritis (RA) and ankylosing spondylitis (AS). As control of inflammation, and hence of symptoms of disease, is increasingly achieved, more attention is given towards the long-term consequences of these disorders, to the structural damage in the skeletal tissues and to the resulting disability. In AS, bone remodeling with new cartilage and bone formation leading to ankylosis is a striking feature. Clinically successful TNF antagonists do not inhibit radiographic progression of disease. New insights into the molecules involved in ankylosis (such as bone morphogenetic proteins and Wnts) have suggested that the classical paradigm linking inflammation and ankylosis can be challenged, and new concepts of disease onset and progression, with a focus on cell stress and damage, are rapidly evolving. In RA, inhibition of Wnt signaling and defective osteoblast function have been associated with lack of repair. As restoration of tissue integrity and homeostasis is the ultimate goal of therapy, these findings suggest new roads for therapeutic intervention. For patients with AS or RA, such strategies will be critically dependent on further research that defines individual risk factors and need for interventions.
Key Points
-
Ankylosing spondylitis (AS) is characterized by extensive cartilage and bone formation, which leads to ankylosis in the sacroiliac joints and the spine
-
Ankylosis and ongoing inflammation both contribute to reduced mobility and increased disability in patients with AS
-
Radiographic progression of disease in AS (characterized by ankylosis) varies greatly between patients
-
In rheumatoid arthritis (RA), suppression of osteoblast activity interferes with healing of erosions; why this inhibition persists even after clinical control of the disease has been achieved is unclear
-
The osteoimmunology research agenda in AS and RA is rapidly evolving, providing new insights into disease onset and the reason why symptoms manifest in the joint or spine
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Klareskog, L., Catrina, A. I. & Paget, S. Rheumatoid arthritis. Lancet 373, 659–672 (2009).
Braun, J. & Sieper, J. Ankylosing spondylitis. Lancet 369, 1379–1390 (2007).
Mease, P. J. Psoriatic arthritis: update on pathophysiology, assessment and management. Ann. Rheum. Dis. 70 (Suppl. 1), i77–i84 (2011).
McInnes, I. B. & O'Dell, J. R. State-of-the-art: rheumatoid arthritis. Ann. Rheum. Dis. 69, 1898–1906 (2010).
Brandt, H. C., Spiller, I., Song, I.-H., Vahldiek, J. L., Rudwaleit, M. & Sieper, J. Performance of referral recommendations in patients with chronic back pain and suspected axial spondyloarthritis. Ann. Rheum. Dis. 66, 1479–1484 (2007).
Luyten, F. P., Lories, R. J. U., Verschueren, P., de Vlam, K. & Westhovens, R. Contemporary concepts of inflammation, damage and repair in rheumatic diseases. Best Pract. Res. Clin. Rheumatol. 20, 829–848 (2006).
Schett, G. Joint remodelling in inflammatory disease. Ann. Rheum. Dis. 66 (Suppl. 3), iii42–iii44 (2007).
Lories, R. J. U., Luyten, F. P. & de Vlam, K. Progress in spondylarthritis. Mechanisms of new bone formation in spondyloarthritis. Arthritis Res. Ther. 11, 221 (2009).
El-Maghraoui, A. Osteoporosis and ankylosing spondylitis. Joint Bone Spine 71, 291–295 (2004).
Carter, S. Lories, R. J. Osteoporosis: a paradox in ankylosing spondylitis. Current Osteoporos. Rep. 9, 112–115 (2011).
Lories, R. J. U., de Vlam, K. & Luyten, F. P. Are current available therapies disease-modifying in spondyloarthritis?. Best. Pract. Res. Clin. Rheumatol. 24, 625–635 (2010).
Machado, P. et al. Both structural damage and inflammation of the spine contribute to impairment of spinal mobility in patients with ankylosing spondylitis. Ann. Rheum. Dis. 69, 1465–1470 (2010).
Vander Cruyssen, B. et al. Hip involvement in ankylosing spondylitis: epidemiology and risk factors associated with hip replacement surgery. Rheumatology (Oxford) 49, 73–81 (2010).
Vandooren, B. et al. Mediators of structural remodeling in peripheral spondylarthritis. Arthritis Rheum. 60, 3534–3545 (2009).
Cross, M. J., Smith, E. U. R., Zochling, J. & March, L. M. Differences and similarities between ankylosing spondylitis and rheumatoid arthritis: epidemiology. Clin. Exp. Rheumatol. 27, S36–S42 (2009).
Boonen, A. & Mau, W. The economic burden of disease: comparison between rheumatoid arthritis and ankylosing spondylitis. Clin. Exp. Rheumatol. 27, S112–S117 (2009).
Sieper, J., Appel, H., Braun, J. & Rudwaleit, M. Critical appraisal of assessment of structural damage in ankylosing spondylitis: implications for treatment outcomes. Arthritis Rheum. 58, 649–656 (2008).
Lories, R. J. U., Derese, I. & Luyten, F. P. Inhibition of osteoclasts does not prevent joint ankylosis in a mouse model of spondyloarthritis. Rheumatology (Oxford) 47, 605–608 (2008).
Schett, G. et al. Tumor necrosis factor α and RANKL blockade cannot halt bony spur formation in experimental inflammatory arthritis. Arthritis Rheum. 60, 2644–2654 (2009).
Maksymowych, W. P. Disease modification in ankylosing spondylitis. Nat. Rev. Rheumatol. 6, 75–81 (2010).
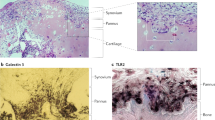
Appel, H. et al. Immunohistochemical analysis of hip arthritis in ankylosing spondylitis: evaluation of the bone-cartilage interface and subchondral bone marrow. Arthritis Rheum. 54, 1805–1813 (2006).
Appel, H. et al. Immunohistochemical analysis of osteoblasts in zygapophyseal joints of patients with ankylosing spondylitis reveal repair mechanisms similar to osteoarthritis. J. Rheumatol. 37, 823–828 (2010).
Bywaters, E. G. The early lesions of ankylosing spondylitis. Ann. Rheum. Dis. 28, 330 (1969).
François, R. J., Gardner, D. L., Degrave, E. J. & Bywaters, E. G. Histopathologic evidence that sacroiliitis in ankylosing spondylitis is not merely enthesitis. Arthritis Rheum. 43, 2011–2024 (2000).
François, R. J., Braun, J. & Khan, M. A. Entheses and enthesitis: a histopathologic review and relevance to spondyloarthritides. Curr. Opin. Rheumatol. 13, 255–264 (2001).
Karsenty, G., Kronenberg, H. M. & Settembre, C. Genetic control of bone formation. Annu. Rev. Cell Dev. Biol. 25, 629–648 (2009).
Lories, R. J. U. & Luyten, F. P. Bone morphogenetic proteins in destructive and remodeling arthritis. Arthritis Res. Ther. 9, 207 (2007).
Lories, R. J. U., Matthys, P., de Vlam, K., Derese, I. & Luyten, F. P. Ankylosing enthesitis, dactylitis, and onychoperiostitis in male DBA/1 mice: a model of psoriatic arthritis. Ann. Rheum. Dis. 63, 595–598 (2004).
Zimmerman, L. B., De Jesús-Escobar, J. M. Harland, R. M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Lories, R. J. U., Derese, I. & Luyten, F. P. Modulation of bone morphogenetic protein signaling inhibits the onset and progression of ankylosing enthesitis. J. Clin. Invest. 115, 1571–1579 (2005).
Lories, R. J. U. et al. Noggin haploinsufficiency differentially affects tissue responses in destructive and remodeling arthritis. Arthritis Rheum. 54, 1736–1746 (2006).
Milat, F. & Ng, K. W. Is Wnt signalling the final common pathway leading to bone formation? Mol. Cell. Endocrinol. 310, 52–62 (2009).
Liu, F., Kohlmeier, S. & Wang, C.-Y. Wnt signaling and skeletal development. Cell Signal 20, 999–1009 (2008).
Lories, R. J. & Luyten, F. P. Osteoimmunology: Wnt antagonists: for better or worse? Nat. Rev. Rheumatol. 5, 420–421 (2009).
Luyten, F. P., Tylzanowski, P. & Lories, R. J. Wnt signaling and osteoarthritis. Bone 44, 522–527 (2009).
Diarra, D. et al. Dickkopf-1 is a master regulator of joint remodeling. Nat. Med. 13, 156–163 (2007).
Uderhardt, S. et al. Blockade of Dickkopf (DKK)-1 induces fusion of sacroiliac joints. Ann. Rheum. Dis. 69, 592–597 (2010).
van Beuningen, H. M., Glansbeek, H. L., van der Kraan, P. M. & van den Berg, W. B. Differential effects of local application of BMP-2 or TGF-β1 on both articular cartilage composition and osteophyte formation. Osteoarthritis Cartilage 6, 306–317 (1998).
Scharstuhl, A., Vitters, E. L., van der Kraan, P. M. & van den Berg, W. B. Reduction of osteophyte formation and synovial thickening by adenoviral overexpression of transforming growth factor β/bone morphogenetic protein inhibitors during experimental osteoarthritis. Arthritis Rheum. 48, 3442–3451 (2003).
Lories, R. J. U., Derese, I., de Bari, C. & Luyten, F. P. Evidence for uncoupling of inflammation and joint remodeling in a mouse model of spondylarthritis. Arthritis Rheum. 56, 489–497 (2007).
van der Heijde, D. et al. Radiographic findings following two years of infliximab therapy in patients with ankylosing spondylitis. Arthritis Rheum. 58, 3063–3070 (2008).
van der Heijde, D. et al. Radiographic progression of ankylosing spondylitis after up to two years of treatment with etanercept. Arthritis Rheum. 58, 1324–1331 (2008).
van der Heijde, D. et al. Canadian (M03–606) study group & ATLAS study group Assessment of radiographic progression in the spines of patients with ankylosing spondylitis treated with adalimumab for up to 2 years. Arthritis Res. Ther. 11, R127 (2009).
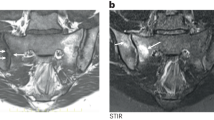
Braun, J. & Baraliakos, X. Imaging of axial spondyloarthritis including ankylosing spondylitis. Ann. Rheum. Dis. 70 (Suppl. 1), i97–i103 (2011).
Spoorenberg, A. et al. Measuring disease activity in ankylosing spondylitis: patient and physician have different perspectives. Rheumatology (Oxford) 44, 789–795 (2005).
Maksymowych, W. P. Progress in spondylarthritis. Spondyloarthritis: lessons from imaging. Arthritis Res. Ther. 11, 222 (2009).
Sieper, J. et al. Persistent reduction of spinal inflammation as assessed by magnetic resonance imaging in patients with ankylosing spondylitis after 2 yrs of treatment with the anti-tumour necrosis factor agent infliximab. Rheumatology (Oxford) 44, 1525–1530 (2005).
Maksymowych, W. P. et al. Inflammatory lesions of the spine on magnetic resonance imaging predict the development of new syndesmophytes in ankylosing spondylitis: evidence of a relationship between inflammation and new bone formation. Arthritis Rheum. 60, 93–102 (2009).
Lu, X. et al. Identification of the homeobox protein Prx1 (MHox, Prrx-1) as a regulator of osterix expression and mediator of tumor necrosis factor α action in osteoblast differentiation. J. Bone Miner. Res. 26, 209–219 (2011).
Kontoyiannis, D., Pasparakis, M., Pizarro, T. T., Cominelli, F. & Kollias, G. Impaired on/off regulation of TNF biosynthesis in mice lacking TNF AU-rich elements: implications for joint and gut-associated immunopathologies. Immunity 10, 387–398 (1999).
Kaplan, F. S. et al. Fibrodysplasia ossificans progressiva. Best Pract. Res. Clin. Rheumatol. 22, 191–205 (2008).
Shore, E. M. et al. A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nat. Genet. 38, 525–527 (2006).
Fukuda, T. et al. Generation of a mouse with conditionally activated signaling through the BMP receptor, ALK2. Genesis 44, 159–167 (2006).
Yu, P. B. et al. BMP type I receptor inhibition reduces heterotopic ossification. Nat. Med. 14, 1363–1369 (2008).
Colbert, R. A., DeLay, M. L., Klenk, E. I. & Layh-Schmitt, G. From HLA-B27 to spondyloarthritis: a journey through the ER. Immunol. Rev. 233, 181–202 (2010).
Benjamin, M. & McGonagle, D. The enthesis organ concept and its relevance to the spondyloarthropathies. Adv. Exp. Med. Biol. 649, 57–70 (2009).
McGonagle, D., Lories, R. J. U., Tan, A. L. & Benjamin, M. The concept of a “synovio-entheseal complex” and its implications for understanding joint inflammation and damage in psoriatic arthritis and beyond. Arthritis Rheum. 56, 2482–2491 (2007).
Baraliakos, X., Listing, J., von der Recke, A. & Braun, J. The natural course of radiographic progression in ankylosing spondylitis—evidence for major individual variations in a large proportion of patients. J. Rheumatol. 36, 997–1002 (2009).
Chen, H. A. et al. Association of bone morphogenetic proteins with spinal fusion in ankylosing spondylitis. J. Rheumatol. 37, 2126–2132 (2010).
Tsukahara, S. et al. COL6A1, the candidate gene for ossification of the posterior longitudinal ligament, is associated with diffuse idiopathic skeletal hyperostosis in Japanese. Spine (Phila Pa 1976) 30, 2321–2324 (2005)
Thomas, G. P. & Brown, M. A. Genetics and genomics of ankylosing spondylitis. Immunol. Rev. 233, 162–180 (2010).
Wanders, A. et al. Nonsteroidal antiinflammatory drugs reduce radiographic progression in patients with ankylosing spondylitis: a randomized clinical trial. Arthritis Rheum. 52, 1756–1765 (2005).
Daoussis, D. et al. Evidence that Dkk-1 is dysfunctional in ankylosing spondylitis. Arthritis Rheum. 62, 150–158 (2010).
Li, X. et al. Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J. Biol. Chem. 280, 19883–19887 (2005).
Appel, H. et al. Altered skeletal expression of sclerostin and its link to radiographic progression in ankylosing spondylitis. Arthritis Rheum. 60, 3257–3262 (2009).
Herman, S., Krönke, G. & Schett, G. Molecular mechanisms of inflammatory bone damage: emerging targets for therapy. Trends Mol. Med. 14, 245–253 (2008).
Bartok, B. & Firestein, G. S. Fibroblast-like synoviocytes: key effector cells in rheumatoid arthritis. Immunol. Rev. 233, 233–255 (2010).
Parsonage, G., Filer, A. D., Haworth, O., Nash, G. B., Rainger, G. E., Salmon, M. & Buckley, C. D. A stromal address code defined by fibroblasts. Trends Immunol. 26, 150–156 (2005).
Walsh, N. C. & Gravallese, E. M. Bone remodeling in rheumatic disease: a question of balance. Immunol. Rev. 233, 301–312 (2010).
Schett, G. & David, J.-P. The multiple faces of autoimmune-mediated bone loss. Nat. Rev. Endocrinol. 6, 698–706 (2010).
Cohen, S. B. et al. Denosumab treatment effects on structural damage, bone mineral density, and bone turnover in rheumatoid arthritis: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, phase II clinical trial. Arthritis Rheum. 58, 1299–1309 (2008).
Cummings, S. R. et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 361, 756–765 (2009).
Scott, D. L., Wolfe, F. & Huizinga, T. W. J. Rheumatoid arthritis. Lancet 376, 1094–1108 (2010).
Døhn, U. M. et al. No overall progression and occasional repair of erosions despite persistent inflammation in adalimumab-treated rheumatoid arthritis patients: results from a longitudinal comparative MRI, ultrasonography, CT and radiography study. Ann. Rheum. Dis. 70, 252–258 (2011).
Walsh, N. C. et al. Osteoblast function is compromised at sites of focal bone erosion in inflammatory arthritis. J. Bone Miner. Res. 24, 1572–1585 (2009).
Aletaha, D., Funovits, J. & Smolen, J. S. Physical disability in rheumatoid arthritis is associated with cartilage damage rather than bone destruction. Ann. Rheum. Dis. 70, 733–739 (2011).
Benjamin, M. & McGonagle, D. The anatomical basis for disease localisation in seronegative spondyloarthropathy at entheses and related sites. J. Anat. 199, 503–526 (2001).
Sieber, C., Kopf, J., Hiepen, C. & Knaus, P. Recent advances in BMP receptor signaling. Cytokine Growth Factor Rev. 20, 343–355 (2009).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author has acted as a consultant for Abbott, MSD and Pfizer, has been a member of speakers bureau for MSD and Pfizer, and received grant or research support from Abbott and Pfizer. Katholieke Universiteit Leuven, Belgium, holds a patent on behalf of the author related to the use of bone morphogenetic protein antagonists in spondyloarthritis.
Rights and permissions
About this article
Cite this article
Lories, R. The balance of tissue repair and remodeling in chronic arthritis. Nat Rev Rheumatol 7, 700–707 (2011). https://doi.org/10.1038/nrrheum.2011.156
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.156
This article is cited by
-
Transfer of microRNA-22-3p by M2 macrophage-derived extracellular vesicles facilitates the development of ankylosing spondylitis through the PER2-mediated Wnt/β-catenin axis
Cell Death Discovery (2022)
-
Targeting chondrocytes for arresting bony fusion in ankylosing spondylitis
Nature Communications (2021)
-
Enhanced osteogenic differentiation of mesenchymal stem cells in ankylosing spondylitis: a study based on a three-dimensional biomimetic environment
Cell Death & Disease (2019)
-
Anti-SIRT1 autoantibody is elevated in ankylosing spondylitis: a potential disease biomarker
BMC Immunology (2018)
-
Bone Disease in Axial Spondyloarthritis
Calcified Tissue International (2018)