Abstract
Antagonists of tumor necrosis factor (TNF) have revolutionized the treatment of selected inflammatory diseases. In rheumatology, this has been most notable for ankylosing spondylitis, psoriatic arthritis and rheumatoid arthritis. Despite their specificity for TNF, these agents, which include the soluble p75 receptor etanercept and the anti-TNF antibodies adalimumab and infliximab, have demonstrated differential clinical efficacy in studies of rheumatoid arthritis; patients who do not respond to one antagonist often respond to another. Therapeutic disparity of these agents is also seen in specific diseases, most notably Crohn's disease. Differences in pharmacodynamics, pharmacokinetics and mechanisms of action, as well as disease heterogeneity, have been proposed to account for these effects. Reverse signaling by transmembrane TNF in response to anti-TNF antibodies, but not soluble receptor, might also influence the therapeutic response.
Key Points
-
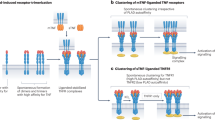
Tumor necrosis factor (TNF) exerts its pleiotropic effects through soluble and transmembrane forms, which interact with two different forms of the receptor—p55 and p75, respectively
-
The remarkable efficacy of TNF antagonists on inflammation is induced through apoptosis-dependent and apoptosis-independent effects that might vary in an organ-specific and disease-specific manner
-
TNF antagonist efficacy is influenced by differences in pharmacodynamics, ligand specificity, and qualitative and quantitative interactions with soluble and transmembrane TNF
-
Clarification of the mechanism of action of TNF antagonists in humans will require agents that selectively antagonize specific forms of TNF or block its actions through monomeric interactions
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Feldmann M et al. (2005) Anti-TNF therapy: where have we got to in 2005? J Autoimmun 25 (Suppl): 26–28
Ritchlin CT and Daikh BE (2001) Recent advances in the treatment of the seronegative spondyloarthropathies. Curr Rheumatol Rep 3: 399–403
Beutler B and Cerami A (1989) The biology of cachectin/TNF—a primary mediator of the host response. Annu Rev Immunol 7: 625–655
Grell M et al. (1995) The transmembrane form of tumor necrosis factor is the prime activating ligand of the 80 kDa tumor necrosis factor receptor. Cell 83: 793–802
Kontoyiannis D et al. (1999) Impaired on/off regulation of TNF biosynthesis in mice lacking TNF AU-rich elements: implications for joint and gut-associated immunopathologies. Immunity 10: 387–398
Zalevsky J et al. (2006) Preclinical efficacy of Xpro 1595, a biologic dominant negative inhibitor of soluble TNF that blocks inflammation without suppressing innate immunity [abstract]. Arthritis Rheum 54 (Suppl): S179
Alexopoulou L et al. (2006) Transmembrane TNF protects mutant mice against intracellular bacterial infections, chronic inflammation and autoimmunity. Eur J Immunol 36: 2768–2780
Ehlers S et al. (2000) Lethal granuloma disintegration in mycobacteria-infected TNFRp55−/− mice is dependent on T cells and IL-12. J Immunol 165: 483–492
Saunders BM et al. (2005) Transmembrane TNF is sufficient to initiate cell migration and granuloma formation and provide acute, but not long-term, control of Mycobacterium tuberculosis infection. J Immunol 174: 4852–4859
Baud V and Karin M (2001) Signal transduction by tumor necrosis factor and its relatives. Trends Cell Biol 11: 372–377
Sandborn WJ et al. (2001) Etanercept for active Crohn's disease: a randomized, double-blind, placebo-controlled trial. Gastroenterology 121: 1088–1094
ten Hove T et al. (2002) Infliximab treatment induces apoptosis of lamina propria T lymphocytes in Crohn's disease. Gut 50: 206–211
Papadakis KA et al. (2005) Safety and efficacy of adalimumab (D2E7) in Crohn's disease patients with an attenuated response to infliximab. Am J Gastroenterol 100: 75–79
Wegener's Granulomatosis Etanercept Trial (WGET) Research Group (2005) Etanercept plus standard therapy for Wegener's granulomatosis. N Engl J Med 352: 351–361
Utz JP et al. (2003) Etanercept for the treatment of stage II and III progressive pulmonary sarcoidosis. Chest 124: 177–185
Lamprecht P et al. (2002) Effectiveness of TNF-α blockade with infliximab in refractory Wegener's granulomatosis. Rheumatology (Oxford) 41: 1303–1307
Serio RN (2003) Infliximab treatment of sarcoidosis. Ann Pharmacother 37: 577–581
Guignard S et al. (2006) Efficacy of tumour necrosis factor blockers in reducing uveitis flares in patients with spondylarthropathy: a retrospective study. Ann Rheum Dis 65: 1631–1634
Wallis RS et al. (2004) Granulomatous infectious diseases associated with tumor necrosis factor antagonists. Clin Infect Dis 38: 1261–1265
Wallis RS et al. (2004) A study of the safety, immunology, virology, and microbiology of adjunctive etanercept in HIV-1-associated tuberculosis. AIDS 18: 257–264
Nestorov I (2005) Clinical pharmacokinetics of TNF antagonists: how do they differ? Semin Arthritis Rheum 34 (Suppl 1): 12–18
Van den Brande JM et al. (2003) Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn's disease. Gastroenterology 124: 1774–1785
Buch MH et al. (2004) True infliximab resistance in rheumatoid arthritis: a role for lymphotoxin alpha? Ann Rheum Dis 63: 1344–1346
Scallon B et al. (2002) Binding and functional comparisons of two types of tumor necrosis factor antagonists. J Pharmacol Exp Ther 301: 418–426
Kohno T et al. (2005) Adalimumab and infliximab bind to Fc-receptors and C1q and generate immunoprecipitation: a differential mechanism from etanercept. Arthritis Rheum 52 (Suppl): S562–S563
Santora LC et al. (2001) Characterization of noncovalent complexes of recombinant human monoclonal antibody and antigen using cation exchange, size exclusion chromatography, and BIAcore. Anal Biochem 299: 119–129
Watts AD et al. (1999) A casein kinase I motif present in the cytoplasmic domain of members of the tumour necrosis factor ligand family is implicated in 'reverse signalling'. EMBO J 18: 2119–2126
Mitoma H et al. (2005) Infliximab induces potent anti-inflammatory responses by outside-to-inside signals through transmembrane TNF-alpha. Gastroenterology 128: 376–392
Smeets TJ et al. (2003) Tumor necrosis factor alpha blockade reduces the synovial cell infiltrate early after initiation of treatment, but apparently not by induction of apoptosis in synovial tissue. Arthritis Rheum 48: 2155–2162
Goedkoop AY et al. (2004) Early effects of tumour necrosis factor alpha blockade on skin and synovial tissue in patients with active psoriasis and psoriatic arthritis. Ann Rheum Dis 63: 769–773
Catrina AI et al. (2005) Evidence that anti-tumor necrosis factor therapy with both etanercept and infliximab induces apoptosis in macrophages, but not lymphocytes, in rheumatoid arthritis joints: extended report. Arthritis Rheum 52: 61–72
Schreiber S et al. (2005) A randomized, placebo-controlled trial of certolizumab pegol (CDP870) for treatment of Crohn's disease. Gastroenterology 129: 807–818
Sandborn WJ et al. Certolizumab pegol, a humanized anti-TNF PEGylated Fab' fragment, is effective and well tolerated in the maintenance of response and remission following induction therapy in active Crohn's disease: a Phase III study (PRECiSE 2). In American College of Gastroenterology 70th annual meeting, 2005 October 28–November 2, Honolulu
Acknowledgements
I thank the careful reading of the manuscript and suggestions by P-P Tak and J Yost.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Rigby, W. Drug Insight: different mechanisms of action of tumor necrosis factor antagonists—passive-aggressive behavior?. Nat Rev Rheumatol 3, 227–233 (2007). https://doi.org/10.1038/ncprheum0438
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0438
This article is cited by
-
Fungal Infections in the Era of Biologic Therapies
Current Clinical Microbiology Reports (2015)
-
Novel Mechanisms of Action of the Biologicals in Rheumatic Diseases
Clinical Reviews in Allergy & Immunology (2014)
-
Covalently dimerized Camelidae antihuman TNFa single-domain antibodies expressed in yeast Pichia pastoris show superior neutralizing activity
Applied Microbiology and Biotechnology (2013)
-
Harnessing programmed cell death as a therapeutic strategy in rheumatic diseases
Nature Reviews Rheumatology (2011)
-
TNF-α Antagonists and Immunization
Current Infectious Disease Reports (2011)