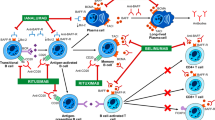
Abstract
As a class, tumor necrosis factor (TNF)-α inhibitors have provided clinicians significant control over chronic inflammatory diseases. With their widespread use has come the emergence of new side effects such as the reactivation of latent infections. One such infection that may reactivate is the hepatitis B virus (HBV). It is currently unknown if HBV reactivation is a class effect or attributable to a particular TNF-α inhibitor. To answer this question, a comprehensive literature review to identify trends in related cases was performed. A systemic literature review was performed using the PubMed and Medline databases (1996 to January 2010) searching for the index term “Hepatitis B” combined with the terms “tumor necrosis factor,” “TNF-α inhibitors,” “etanercept,” “adalimumab,” “certolizumab,” and “golimumab.” All relevant articles in English were reviewed, and secondary references of interest were also retrieved. Thirty-five cases with hepatitis B surface antigen (HBsAg) positivity known prior to initiation of TNF-α inhibitors were identified. Infliximab was used in 17 cases, etanercept in 12 cases, and adalimumab in 6 cases. All six cases of clinically symptomatic hepatitis were associated with infliximab therapy. Infliximab was associated with the most cases of greater than 2-fold increase in alanine aminotransferase (six of nine cases) and greater than 1,000-fold increase in HBV DNA load (three of four). The two deaths reported occurred with infliximab therapy. Potential mechanisms of action for the reported observations include differences in molecular design, route of administration, and potency in clearing TNF-α. In patients with a positive HBsAg prior to starting a TNF-α inhibitor, infliximab has the most reported cases associated with HBV reactivation. While such reactivation may be due to a variety of reasons, clinicians prescribing TNF-α inhibitors to HBsAg-positive patients should consider prophylactic antiviral therapy and close monitoring for any clinical or serological evidence of hepatitis.
Similar content being viewed by others
References
Giles JT, Bathon JM (2004) Serious infections associated with anticytokine therapies in the rheumatic diseases. J Intensive Care Med 19:320–334
Keane J, Gershon S, Wise RP et al (2001) Tuberculosis associated with infliximab, a tumor necrosis factor α-neutralizing agent. N Engl J Med 345:1098–1103
Dienstag JL (2008) Hepatitis B virus infection. N Engl J Med 359(14):1486–1500
Shale MJ, Seow CH, Coffin CS et al (2009) Review article: chronic viral infection in the anti-tumour necrosis factor therapy era in inflammatory bowel disease. Aliment Pharmacol Ther 31:20–34
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2008) Drugs@FDA: Adalimumab Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/125057s114lbl.pdf. Accessed 8 August 2009
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2008) Drugs@FDA: Certolizumab Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/125160s000lbl.pdf. Accessed 8 August 2009
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2008) Drugs@FDA: Etanercept Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/103795s5359lbl.pdf. Accessed 8 August 2009
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2009) Drugs@FDA: Golimumab Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/125289s000lbl.pdf. Accessed 8 August 2009
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2009) Drugs@FDA: Infliximab Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/103772s5234lbl.pdf. Accessed 8 August 2009
Wendling D, Di Martino V, Prati C et al (2009) Spondyloarthropathy and chronic B hepatitis. Effect of anti-TNF therapy. Jt Bone Spine 76:308–311
Wendling D, Auge B, Bettinger D et al (2005) Reactivation of a latent precore mutant hepatitis B virus related chronic hepatitis during infliximab treatment for severe spondyloarthropathy. Ann Rheum Dis 64:788–789
Esteve M, Loras C, González-Huix F (2007) Lamivudine resistance and exacerbation of hepatitis B in infliximab-treated Crohn's disease patient. Inflamm Bowel Dis 13(11):1450–1451
Esteve M, Saro C, González-Huix F et al (2004) Chronic hepatitis B reactivation following infliximab therapy in Crohn's disease patients: need for primary prophylaxis. Gut 53:1363–1365
Zingarelli S, Frassi M, Bazzani C et al (2009) Use of tumor necrosis factor-α-blocking agents in hepatitis B virus-positive patients: reports of 3 cases and review of the literature. J Rheumatol 36(6):1188–1194
Zingarelli S, Airò P, Frassi M et al (2008) Prophylaxis and therapy of HBV infection in 20 patients treated with disease modifying antirheumatic drugs or with biological agents for rheumatic diseases. Reumatismo 60(1):22–27
Roux CH, Brocq O, Breuil V et al (2006) Safety of anti-TNF-α therapy in rheumatoid arthritis and spondyloarthropathies with concurrent B or C chronic hepatitis. Rheumatology 45:1294–1297
Cansu DU, Kalifoglu T, Korkmaz C (2008) Short-term course of chronic hepatitis B and C under treatment with etanercept associated with different disease modifying antirheumatic drugs without antiviral prophylaxis. J Rheumatol 35:421–424
Carroll MB, Bond MI (2008) Use of tumor necrosis factor-α inhibitors in patients with chronic hepatitis B infection. Semin Arthritis Rheum 38(3):208–217
Li S, Kaur PP, Chan V (2009) Use of tumor necrosis factor-α (TNF-α) antagonists infliximab, etanercept, and adalimumab in patients with concurrent rheumatoid arthritis and hepatitis B or hepatitis C: a retrospective record review of 11 patients. Clin Rheumatol 28:787–791
Kaur PP, Chan VC, Berney SN (2008) Histological evaluation of liver in two rheumatoid arthritis patients with chronic hepatitis B and C treated with TNF-alpha blockade: case reports. Clin Rheumatol 27:1069–1071
Millonig G, Kern M, Ludwiczek O et al (2006) Subfulminant hepatitis B after infliximab in Crohn's disease: need for HBV-screening? World J Gastroenterol 12:974–976
Michel M, Duvoux C, Hezode C et al (2003) Fulminant hepatitis after infliximab in a patient with hepatitis B virus treated for an adult onset Still's disease. J Rheumatol 30:1624–1625
Ostuni P, Botsios C, Punzi L et al (2003) Hepatitis B reactivation in a chronic hepatitis B surface antigen carrier with rheumatoid arthritis treated with infliximab and low dose methotrexate. Ann Rheum Dis 62:686–687
Oniankitan O, Duvous C, Challine D et al (2004) Infliximab therapy for rheumatic diseases in patients with chronic hepatitis B or C. J Rheumatol 31:107–109
Ueno Y, Tanaka S, Shimamoto M et al (2005) Infliximab Therapy for Crohn's disease in patient with chronic hepatitis B. Dig Dis Sci 50:163–166
Anelli MG, Torres DD, Manno C et al (2005) Improvement of renal function and disappearance of hepatitis B virus DNA in a patient with rheumatoid arthritis and renal amyloidosis following treatment with infliximab. Arthritis Rheum 52:2519–2520
MdV G-S, Gomez-Camacho F, Poyato-Gonzalez A et al (2004) Infliximab therapy in a patient with Crohn's disease and chronic hepatitis B virus infection. Inflamm Bowel Dis 10(5):701–702
Madonia S, Orlando A, Scimeca D et al (2007) Occult hepatitis B and infliximab-induced HBV reactivation. Inflamm Bowel Dis 13(4):508
Robinson H, Walker-Bone K (2009) Anti-TNF-α therapy for rheumatoid arthritis among patients with chronic hepatitis B infection. Rheumatology 48:450–451
Sakellariou GT, Chatzigiannis I (2007) Long-term anti-TNFα therapy for ankylosing spondylitis in two patients with chronic HBV infection. Clin Rheumatol 26:950–952
Colbert C, Chavarria A, Berkelhammer C (2007) Fulminant hepatic failure in chronic hepatitis B on withdrawal of corticosteroids, azathioprine and infliximab for Crohn's disease. Inflamm Bowel Dis 13:1453–1454
Ojiro K, Naganuma M, Ebinuma H et al (2008) Reactivation of hepatitis B in a patient with Crohn's disease treated using infliximab. J Gastroenterol 43:397–401
Calabrese LH, Zein NN, Vassilopoulos D (2006) Hepatitis B virus (HBV) reactivation with immunosuppressive therapy in rheumatic diseases: assessment and preventive strategies. Ann Rheum Dis 65:983–989
Rehermann B (2000) Intrahepatic T cells in hepatitis B: viral control versus liver cell injury. J Exp Med 191:1263–1268
Cheong JY, Cho SW, Hwang IL et al (2006) Association between chronic hepatitis B virus infection and interleukin-10, tumor necrosis factor-α gene promoter polymorphisms. J Gastro Hepatology 21:1163–1169
Ben-Ari Z, Mor E, Papo O et al (2003) Cytokine gene polymorphisms in patients infected with hepatitis B virus. Am J Gastroenterol 98:144–150
Höhler T, Kruger A, Gerken G et al (1998) A tumour necrosis factor-alpha (TNF-α) promoter polymorphism is associated with chronic hepatitis B infection. Clin Exp Immunol 111:579–582
Li HQ, Li Z, Liu Y et al (2006) Association of-238G/A and -857C/T polymorphisms of tumor necrosis factor-alpha gene promoter region with outcomes of hepatitis B virus infection. Biomed Environ Sci 19(2):133–136
Li HQ, Li Z, Liu Y (2005) Association of polymorphism of tumor necrosis factor-alpha gene promoter region with outcome of hepatitis B virus infection. World J Gastroenterol 11(33):5213–5217
Kim YJ, Lee HS, Yoon JH et al (2003) Association of TNF-alpha promoter polymorphisms with the clearance of hepatitis B virus infection. Hum Mol Genet 12(19):2541–2546
Du T, Guo XH, Zhu XL et al (2006) Association of TNF-alpha promoter polymorphisms with the outcomes of hepatitis B virus infection in Chinese Han population. J Viral Hepat 13(9):618–624
Fang JWS, Shen WW, Meager A et al (1996) Activations of the tumor necrosis factor-α system in the liver in chronic hepatitis B virus infection. Am J Gastroenterol 91:748–753
Rehermann B (2000) Intrahepatic T cells in hepatitis B: viral control versus liver cell injury. J Exp Med 191:1263–1268
Su F, Schneider RJ (1997) Hepatitis B virus HBx protein sensitizes cells to apoptotic killing by tumor necrosis factor α. Proc Natl Acad Sci USA 94:8744–8749
Thimme R, Wieland S, Steiger C et al (2003) CD8+ T cells mediate viral clearance and disease pathogenesis during aucte hepatitis B virus infection. J Virol 77:68–76
Block TM, Guo H, Guo JT (2007) Molecular virology of hepatitis B virus for clinicians. Clin Liver Dis 11:685–706
Petersen J, Lutgehetmann M, Volz T et al (2007) What is the role of cccDNA in chronic HBV infection? Impact on HBV therapy. Hepatol Rev 4:9–13
Gao W, Hu J (2007) Formation of hepatitis B virus covalently closed circular DNA: removal of genome-linked protein. J Virol 81:6164–6174
Sung JJ, Wong ML, Bowden S (2005) Intrahepatic hepatitis B virus covalently closed circular DNA can be a predictor of sustained response to therapy. Gastroenterol 128:1890–1897
Zoulim F (2005) New insight on HBV persistence from the study of intrahepatic viral cccDNA. J Hepatol 42:302–308
Maini MK, Boni C, Lee CK et al (2000) The role of virus-specific CD8+ cells in liver damage and viral control during persistent hepatitis B virus infection. J Exp Med 191:1269–1281
Hussain MJ, Lau JY, Williams R et al (1994) Hepatic expression of tumour necrosis factor-alpha in chronic hepatitis B virus infection. J Clin Pathol 47(12):1112–1115
Ganem D, Prince AM (2004) Hepatitis B virus infection—natural history and clinical consequences. N Engl J Med 350:1118–1129
Jeng JE, Tsai JF, Chuang LY et al (2007) Tumor necrosis factor-alpha 308.2 polymorphism is associated with advanced hepatic fibrosis and higher risk for hepatocellular carcinoma. Neoplasia 9(11):987–992
Remicade website (2010) Centocor. http://www.remicade.com/remicade/global/index.html. Accessed 7 April 2010
Tracey D, Klareskog L, Sasso EH et al (2008) Tumor necrosis factor antagonist mechanisms of action: a comprehensive review. Pharmacol Ther 117:244–279
Furst DE, Beenhouwer O (2006) Tumor necrosis factor antagonists: different kinetics and/or mechanisms of action may explain differences in the risk for developing granulomatous infection. Semin Arthritis Rheum 36(3):159–167
Weaver AL (2003) Differentiating the new rheumatoid arthritis biologic therapies. J Clin Rheumatol 9(2):99–114
Olsen NJ, Stein CM (2004) New drugs for rheumatoid arthritis. N Engl J Med 350(21):2167–2179
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2004) Drugs@FDA: Infliximab Label Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2004/103772_5077_lbl.pdf. Accessed 8 August 2009
Bourne T, Fossati G, Nesbitt A (2008) A PEGylated Fab' fragment against tumor necrosis factor for the treatment of Crohn's disease. Biodrugs 22(5):331–337
Mitoma H, Horiuchi T, Tsukamoto H et al (2008) Mechanisms for cytotoxic effects of anti-tumor necrosis factor agents on transmembrane tumor necrosis factor α-expressing cells: comparison among infliximab, etanercept, and adalimumab. Arthritis Rheum 58(5):1248–1257
Scott DL, Cope A (2009) New tumor necrosis factor inhibitors for rheumatoid arthritis: are there benefits from extending choice? Ann Rheum Dis 68(6):767–769
Fossati G, Nesbitt A (2005) In vitro complement-dependent cytotoxicity and antibody-dependent cellular cytotoxicity by the anti-TNF agents adalimumab, etanercept, infliximab, and certolizumab pegol (CDP870): 807. Am J Gastro 100(supp 1):S299
Saliu OY, Sofer C, Stein D et al (2006) Tumor-necrosis-factor blockers: differential effects on mycobacterial immunity. J Infect Dis 194:486–492
Tubach F, Salmon D, Ravaud P et al (2009) Risk of tuberculosis is higher with anti-tumor necrosis factor monoclonal antibody therapy than with soluble tumor necrosis factor receptor therapy. Arthritis Rheum 60(7):1884–1894
Suwannalai P, Auethavekiat P, Udomsubpayakul U et al (2009) The infectious profiles of anti-tumor necrosis factor agents in a Thai population: a retrospective study a the university-based hospital. Int J Rheum Dis 12(2):118–124
Takeuchi T, Tatsuki Y, Nogami Y et al (2008) Postmarketing surveillance of the safety profile of infliximab in 5000 Japanese patients with rheumatoid arthritis. Ann Rheum Dis 67(2):189–194
Koike T, Harigai M, Inokuma S et al (2009) Postmarketing surveillance of the safety and effectiveness of etanercept in Japan. J Rheum 36(5):898–906
U.S. Food and Drug Administration Center for Drug Evaluation and Research (1999) Drugs@FDA: Infliximab Product Approval Information. <http://www.accessdata.fda.gov/drugsatfda_docs/appletter/1999/inflcen111099L.htm. Accessed 17 March 2010
U.S. Food and Drug Administration Center for Drug Evaluation and Research (2002) Drugs@FDA: Etanercept Product Approval Information-Licensing Action. http://www.accessdata.fda.gov/drugsatfda_docs/appletter/2002/etanimm011502L.htm. Accessed 17 March 2010
Nathan DM, Angus PW, Gibson PR (2006) Hepatitis B and C virus infections and anti-tumor necrosis factor-α therapy: guidelines for clinical approach. J Gastroenterol Hepatol 21:1366–1371
Liang TJ (2009) Hepatitis B: the virus and disease. Hepatology 49(5 Suppl):S13–S21
Saag KG, Teng GG, Patkar NM et al (2008) American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis. Arthritis Rheum 59(6):762–784
Ledingham J, Deighton C (2005) Update on the British Society for Rheumatology guidelines for prescribing TNFalpha blockers in adults with rheumatoid arthritis (update of previous guidelines of April 2001). Rheumatology 44(2):157–163
British Society of Rheumatology. “BSR Guidelines.” http://www.rheumatology.org.uk/guidelines/. Accessed 15 August 2009
Lichtenstein GR, Abreu MT, Cohen R et al (2006) American Gastroenterologic Association Institute medical position statement on corticosteroids, immunomodulators, and infliximab in inflammatory bowel disease. Gastroenterology 130:935–939
Carter MJ, Lobo AJ, Travis SPL (2004) Guidelines for the management of inflammatory bowel disease in adults. Gut 53(Supp V):v1–v16
Desai SB, Furst DE (2006) Problems encountered during anti-tumor necrosis factor therapy. Best Prac & Res Clin Rheum 20(4):757–790
Vassilopoulos D, Calabrese LH (2007) Risks of immunosuppressive therapies including biologic agents in patients with rheumatic diseases and co-existing chronic viral infections. Curr Opin Rheumatol 19:619–625
Marignani M, Cox MC, Delle Fave G (2009) Hepatitis B virus infection. N Engl J Med 360(3):304–306
Tilg H, Kaser A, Moschen AR (2006) How to modulate inflammatory cytokines in liver disease. Liver Int 26:1029–1039
Calabrese LH, Zein NN, Vassilopoulos D (2006) Hepatitis B virus (HBV) reactivation with immunosuppressive therapy in rheumatic diseases: assessment and preventive strategies. Ann Rheum Dis 65:983–989
Cheong JY, Cho SW, Hwang IL et al (2006) Association between chronic hepatitis B virus infection and interleukin-10, tumor necrosis factor-α gene promoter polymorphisms. J Gastro Hepatology 21:1163–1169
Thio CL, Thomas DL, Carrington M (2000) Chronic viral hepatitis and the human genome. Hepatology 31:819–827
Yang HI, Lu SN, Liaw YF et al (2002) Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med 347(3):168–174
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors confirm that this manuscript has neither been submitted nor is simultaneously being submitted elsewhere. No portion of the data has been or will be published in proceedings or transactions of meetings or symposium volumes.
The authors have no financial support or other benefits from commercial sources to disclose. They received no pharmaceutical or industry support in writing this manuscript.
The views expressed in this article are those of the authors and do not reflect the official policy or position of the United States Air Force, Department of Defense, or the US Government.
Rights and permissions
About this article
Cite this article
Carroll, M.B., Forgione, M.A. Use of tumor necrosis factor α inhibitors in hepatitis B surface antigen-positive patients: a literature review and potential mechanisms of action. Clin Rheumatol 29, 1021–1029 (2010). https://doi.org/10.1007/s10067-010-1523-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-010-1523-2