Abstract
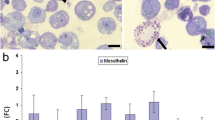
Inflammatory and neoplastic disease processes of the abdominal cavity are frequently associated with disruption of the integrity of the peritoneal mesothelium. In the present study, we analyzed the effects of the pro-inflammatory cytokines interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) on the morphology and expression of adhesion molecules of human peritoneal mesothelial cells (HPMC). Treatment of HPMC with IL-1β and TNF-α resulted in a time- and dose-dependent alteration of the normal cobblestone morphology of the mesothelium with loss of polarization, cellular retraction and exposure of the submesothelial matrix. The effect was already observable after 6 h of treatment and was most pronounced at a dose of 10 ng/ml of IL-1β or TNF-α. These morphological alterations were associated with a significant rearrangement of the expression of mesothelial adhesion molecules as detected by flow cytometry. IL-1β and TNF-α both led to a loss of the expression of the hemidesmosomal integrin subunits α6 (P<0.01 and P<0.001) and β4 (P<0.01) and an increased expression of the integrin subunit α5 (P<0.001 and P<0.01). IL-1β furthermore upregulated the expression of the integrin subunits α1, α2 and the adhesion molecule CD44 while the latter was downregulated by TNF-α. Our data indicate that IL-1β and TNF-α may significantly affect disease processes of the abdominal cavity by their potential to disrupt the mesothelial basal cell-matrix adhesion and, thus, the integrity of the peritoneal mesothelial cell lining.



Similar content being viewed by others
References
Alt A, Ohba M, Li L, Gartsbein M, Belanger A, Denning MF, Kuroki T, Yuspa SH, Tennenbaum T (2001) Protein kinase C-delta-mediated phosphorylation of alpha6beta4 is associated with reduced integrin localization to the hemidesmosome and decreased keratinocyte attachment. Cancer Res 61:4591–4598
Amberger A, Maczek C, Jurgens G, Michaelis D, Schett G, Trieb K, Eberl T, Jindal S, Xu Q, Wick G (1997) Co-expression of ICAM-1, VCAM-1, ELAM-1 and Hsp60 in human arterial and venous endothelial cells in response to cytokines and oxidized low-density lipoproteins. Cell Stress Chaperones 2:94–103
Andreoli SP, Mallett C, Williams K, McAteer JA, Rothlein R, Doerschuk CM (1994) Mechanisms of polymorphonuclear leukocyte mediated peritoneal mesothelial cell injury. Kidney Int 46:1100–1109
Bittinger F, Schepp C, Brochhausen C, Lehr HA, Otto M, Kohler H, Skarke C, Walgenbach S, Kirkpatrick CJ (1999) Remodeling of peritoneal-like structures by mesothelial cells: its role in peritoneal healing. J Surg Res 82:28–33
Borradori L, Sonnenberg A (1999) Structure and function of hemidesmosomes: more than simple adhesion complexes. J Invest Dermatol 112:411–418
Cannistra SA, Kansas GS, Niloff J, DeFranzo B, Kim Y, Ottensmeier C (1993) Binding of ovarian cancer cells to peritoneal mesothelium in vitro is partly mediated by CD44. Cancer Res 53:3830–3838
Carter D, True L, Otis CN (1997) Serous membranes: histology for pathologists. In: Sternberg SS (ed) Lippincott–Raven, New York, pp 2299–2328
Casey RC, Burleson KM, Skubitz KM, Pambuccian SE, Oegema TR Jr, Ruff LE, Skubitz AP (2001) Beta 1-integrins regulate the formation and adhesion of ovarian carcinoma multicellular spheroids. Am J Pathol 159:2071–2080
Cronauer MV, Stadlmann S, Klocker H, Abendstein B, Eder IE, Rogatsch H, Zeimet AG, Marth C, Offner FA (1999) Basic fibroblast growth factor synthesis by human peritoneal mesothelial cells. Am J Pathol 155:1977–1984
Dowling J, Yu QC, Fuchs E (1996) Beta4 integrin is required for hemidesmosome formation, cell adhesion and cell survival. J Cell Biol 134:559–572
Giancotti FG, Ruoslahti E (1999). Integrin signaling. Science 285:1028–1032
Goodison S, Urquidi V, Tarin D (1999). CD44 cell adhesion molecules. Mol Pathol 52:189–196
Hosono J, Narita T, Kimura N, Sato M, Nakashio T, Kasai Y, Nonami T, Nakao A, Takagi H, Kannagi R (1998) Involvement of adhesion molecules in metastasis of SW1990, human pancreatic cancer cells. J Surg Oncol 67:77–84
Ikubo A, Morisaki T, Katano M, Kitsuki H, Anan K, Uchiyama A, Tanaka M, Torisu M (1995) A possible role of TGF-beta in the formation of malignant effusions. Clin Immunol Immunopathol 77:27–32
Klaus A, Margreiter R, Pernthaler H, Klima G, Offner FA (2003) Diffuse mesenterial sclerosis: a characteristic feature of chronic small-bowel allograft rejection. Virchows Arch 442:48–55
Kutteh WH, Kutteh CC (1992) Quantitation of tumor necrosis factor-alpha, interleukin-1 beta, and interleukin-6 in the effusions of ovarian epithelial neoplasms. Am J Obstet Gynecol 167:1864–1869
Lessan K, Aguiar DJ, Oegema T, Siebenson L, Skubitz AP (1999) CD44 and beta1 integrin mediate ovarian carcinoma cell adhesion to peritoneal mesothelial cells. Am J Pathol 154:1525–1537
Li FK, Davenport A, Robson RL, Loetscher P, Rothlein R, Williams JD, Topley N (1998) Leukocyte migration across human peritoneal mesothelial cells is dependent on directed chemokine secretion and ICAM-1 expression. Kidney Int 54:2170–2183
Mandl-Weber S, Cohen CD, Haslinger B, Kretzler M, Sitter T (2002) Vascular endothelial growth factor production and regulation in human peritoneal mesothelial cells. Kidney Int 61:570–578
Margetts PJ, Kolb M, Yu L, Hoff CM, Holmes CJ, Anthony DC, Gauldie J (2002) Inflammatory cytokines, angiogenesis, and fibrosis in the rat peritoneum. Am J Pathol 160:2285–2294
Nagy JA, Morgan ES, Herzberg KT, Manseau EJ, Dvorak AM, Dvorak HF (1995) Pathogenesis of ascites tumor growth: angiogenesis, vascular remodeling and stroma formation in the peritoneal lining. Cancer Res 55:376–385
Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C (1991) A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Methods 139:271–279
Nishimura S, Chung YS, Yashiro M, Inoue T, Sowa M (1996) CD44H plays an important role in peritoneal dissemination of scirrhous gastric cancer cells. Jpn J Cancer Res 87:1235–1244
Offner FA, Obrist P, Stadlmann S, Feichtinger H, Klingler P, Herold M, Zwierzina H, Hittmair A, Mikuz G, Abendstein B, Zeimet A, Marth C (1995) IL-6 secretion by human peritoneal mesothelial and ovarian cancer cells. Cytokine 7:542–547
Offner FA, Feichtinger H, Stadlmann S, Obrist P, Marth C, Klingler P, Grage B, Schmahl M, Knabbe C (1996) Transforming growth factor-β synthesis by human peritoneal mesothelial cells. Induction by interleukin-1. Am J Pathol 148:1679–1688
Propst T, Propst A, Herold M, Schauer G, Judmaier G, Braunsteiner H, Stoffler G, Vogel W (1993) Spontaneous bacterial peritonitis is associated with high levels of interleukin-6 and its secondary mediators in ascitic fluid. Eur J Clin Invest 23:832–836
Runyon BA, Hillebrand DJ (1998) Surgical peritonitis and other diseases of the peritoneum, mesentery, momentum, and diaphragm. In: Feldman M, Scharschmidt BF, Sleisenger MH (eds) Gastrointestinal and liver disease. Pathophysiology, diagnosis, management. Saunders, Philadelphia, pp 2035–2046
Ruoslahti E, Vaheri A (1997). Cell-to-cell contact and extracellular matrix. Curr Opin Cell Biol 9:605–607
Sachs L (1997) Angewandte statistik. In: Sachs L (ed) Anwendung statistischer methoden. Springer, Berlin Heidelberg New York, pp 335–337
Skubitz AP, Bast RCJr, Wayner EA, Letourneau PC, Wilke MS (1996) Expression of alpha 6 and beta 4 integrins in serous ovarian carcinoma correlates with expression of the basement membrane protein laminin. Am J Pathol 148:1445–1461
Sriram PS, Mohammed KA, Nasreen N, Hardwick J, Van Horn R, Sanders K, Antony VB (2002) Adherence of ovarian cancer cells induces pleural mesothelial cell (PMC) permeability. Oncol Res 13:79–85
Strobel T, Cannistra SA (1999) Beta1-integrins partly mediate binding of ovarian cancer cells to peritoneal mesothelium in vitro. Gynecol Oncol 73:362–367
Tietze L, Borntraeger J, Klosterhalfen B, Amo-Takyi B, Handt S, Gunther K, Merkelbach-Bruse S (1999) Expression and function of beta(1) and beta(3) integrins of human mesothelial cells in vitro. Exp Mol Pathol 66:131–139
Topley N (1995) The cytokine network controlling peritoneal inflammation. Perit Dial Int 15:35–40
Topley N, Joerres A, Luttmann W, Petersen MM, Lang J, Thierauch KH, Muller C, Coles GA, Davies M, Williams JD (1993) Human peritoneal mesothelial cells synthesize interleukin-6: induction by IL-1β and TNFα. Kidney Int 43:226–233
Topley N, Brown Z, Joerres A, Westwick J, Davies M, Coles GA, Williams JD (1993) Human peritoneal mesothelial cells synthesize interleukin-8: synergistic induction by interleukin-1 beta and tumor necrosis factor-alpha. Am J Pathol 142:1876–1886
Van Rossen ME, Hofland LJ, van den Tol MP, van Koetsveld PM, Jeekel J, Marquet RL, van Eijck CH (2001) Effect of inflammatory cytokines and growth factors on tumour cell adhesion to the peritoneum. J Pathol 193:530–537
Volz J, Volz-Koster S, Kanis S, Klee D, Ahlert C, Melchert F (2000) Modulation of tumor-induced lethality after pneumoperitoneum in a mouse model. Cancer 89:262–266
Whitaker D, Papadimitriou J (1985) Mesothelial healing: morphological and kinetic investigations. J Pathol 145:159–175
Witz CA, Montoya-Rodriguez IA, Miller DM, Schneider BG, Schenken RS (1998) Mesothelium expression of integrins in vivo and in vitro. J Soc Gynecol Invest 5:87–93
Zeimet AG, Widschwendter M, Knabbe C, Fuchs D, Herold M, Muller-Holzner E, Daxenbichler G, Offner FA, Dapunt O, Marth C (1998) Ascitic interleukin-12 is an independent prognostic factor in ovarian cancer. J Clin Oncol 16:1861–1868
Acknowledgements
The excellent technical assistance of Mrs. I. Jehart is gratefully acknowledged. We would also like to thank Mr. J. Pollheimer for helpful comments on statistics.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stadlmann, S., Raffeiner, R., Amberger, A. et al. Disruption of the integrity of human peritoneal mesothelium by interleukin-1β and tumor necrosis factor-α. Virchows Arch 443, 678–685 (2003). https://doi.org/10.1007/s00428-003-0867-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-003-0867-2