Abstract
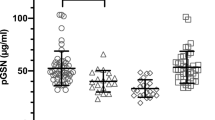
Elevation of circulating phospholipase A2 (PLA2) activity is associated with sepsis and septic shock. Elevated levels of PLA2 activity also are seen in association with chronic inflammatory disorders such as rheumatoid arthritis. The relationship between these phospholipases is unclear. We have developed a highly specific enzyme-linked immunosorbent assay (ELISA) capable of measuring human synovial PLA2 in plasma, using monoclonal antibodies raised to recombinant synovial PLA2. This ELISA has been used to quantitate circulating PLA2 levels in patients clinically diagnosed with sepsis. These elevated levels positively correlated with the elevation seen in plasma PLA2 enzyme activity. The antibodies also have been used to purify immunoreactive PLA2 from plasma of patients with sepsis, thus enabling characterization of the purified protein by amino-terminal sequence analysis. We conclude from this study that the increase in PLA2 activity seen in association with sepsis and septic shock results from a dramatic elevation in levels of a circulating PLA2 enzyme. This inflammatory PLA2 is indistinguishable, both immunologically and chemically, from that associated with rheumatoid arthritis. Therapeutic agents directed towards inhibition of this inflammatory PLA2 enzyme may have utility in the treatment of both chronic and acute inflammatory disease.
Similar content being viewed by others
References
Parker, M. M., andJ. E. Parillo. 1983. Septic shock: Hemodynamics and pathogenesis.JAMA 250:3324–3327.
Shubin, H., andM. H. Weil. 1976. Bacterial shock.JAMA 235:421–424.
Kreger, B. E., D. E. Craven, andW. R. McCabe. 1980. Gram-negative bacteraemia. III. Reassessment of etiology, epidemiology and ecology in 612 patients.Am. J. Med. 63:332–343.
Kreger, B. E., D. E. Craven, andW. R. McCabe. 1980. Gram-negative bacteraemia. IV. Re-evaluation of clinical features and treatment in 612 patients.Am. J. Med. 68:344–355.
MacLean, L. D., W. G. Mulligan, A. P. H. McClean, andJ. H. Duff. 1967. Patterns of septic shock in man: a detailed study of 56 patients.Ann. Surg. 166:543–562.
Pollack, M. M., A. I. Fields, andU. E. Ruttimann. 1985. Distributions of cardiopulmonary variables in pediatric survivors and nonsurvivors of septic shock.Crit. Care. Med. 13:454–459.
Abraham, E., W. C. Shoemaker, R. D. Bland, andJ. C. Cobo. 1983. Sequential cardiorespiratory patterns in septic shock.Crit. Care. Med. 11:799–803.
Abraham, E., R. D. Bland, J. C. Cobo, andW. C. Shoemaker. 1984. Sequential cardiorespiratory patterns associated with outcome in septic shock.Chest 85:75–80.
Fein, A. M., M. Lippmann, H. Holtzman, A. Eliraz, andS. K. Goldberg. 1983. The risk factors, incidence, and prognosis of ARDS following septicaemia.Chest 83:40–42.
Kaplan, R. L., S. A. Sahn, andT. L. Petty. 1979. Incidence and outcome of the respiratory distress syndrome in gram-negative sepsis.Arch. Intern. Med. 139:867–869.
Colucci, M., J. A. Paramo, andD. Collen. 1985. Generation in plasma of a fast-acting inhibitor of plasminogen activator in response to endotoxin stimulation.J. Clin. Invest. 75:818–824.
Morrison, D. C., andJ. L. Ryan. 1987. Endotoxins and disease mechanisms.Annu. Rev. Med. 38:417–432.
Suffredini, A. F., P. C. Harpel, andJ. E. Parillo. 1989. Promotion and subsequent inhibition of plasminogen activation after administration of intravenous endotoxin to normal subjects.N. Engt. J. Med. 320:1165–1172.
Bendtzen, K. 1988. Interleukin 1, interleukin 6 and tumor necrosis factor in infection, inflammation and immunity.Immunol. Lett. 19:183–191.
Beutler, B., andA. Cerami. 1987. Cachectin: more than a tumor necrosis factor.N. Eng. J. Med. 316:379–385.
Clark, M. A., M. J. Chen, S. T. Crooke, andJ. S. Bomalaski. 1988. Tumour necrosis factor (cachectin) induces phospholipase A2 activity and the synthesis of a phospholipase A2activating protein in endothelial cells.Biochem. J. 250:125–132.
Heinrich, P. C., J. V. Castell, andT. Andus. 1990. Interieukin-6 and the acute phase response.Biochem. J. 265:621–636.
Stern, D. M., I. Bank, P. P. Nawroth, J. Cassimeris, W. Kiseil, W. Fenton II, C. Dinarello, L. Chess, andL. Jaffe. 1985. Self-regulation of procoagulant events on the endothelial cell surface.J. Exp. Med. 162:1223–1235.
Coalson, J. J. 1986. Pathology of sepsis, septic shock and multiple organ failure.In Perspective on Sepsis and Septic Shock. W. J. Sibbald and C. L. Sprung, editors. Society of Critical Care Medicine, Fullerton, California:27–59.
McGovern, V. J. 1984. Shock revisited.Pathol. Ann. 19:15–36.
Feuerstein, G., andJ. M. Hallenbeck. 1987. Prostaglandins, leukotrienes, and plateletactivating factor in shock.Annu. Rev. Pharmacol. Toxicol. 27:301–313.
Irvine, R. F. 1982. How is the level of free arachidonic acid controlled in mammalian cells?Biochem. J. 204:3–16.
Slotman, G. J., K. W. Burchard, J. J. Williams, A. D'arezzo, andS. A. Yellin. 1986. Interaction of prostaglandins, activated complement and granulocytes in clinical sepsis and hypotension.Surgery 99:744–750.
Vadas, P., andJ. B. Hay. 1983. Involvement of circulating phospholipase A2 in the pathogenesis of the hemodynamic change in endotoxin shock.Can. J. Physiol. Pharmacol. 61:561–566.
Vadas, P. 1984. Elevated plasma phospholipase A2 levels: Correlation with the hemodynamic and pulmonary changes in gram-negative septic shock in man.J. Lab. Clin. Med. 104:873–881.
Vadas, P., W. Pruzanski, E. Stefanski, B. Sternby, R. Mustard, J. Bohnen, I. Fraser, V. Farewell, andC. Bombardier. 1988. Pathogenesis of hypotension in septic shock: Correlation of circulatory phospholipase A2 levels with circulatory collapse.Crit. Care. Med. 16:1–6.
Seilhamer, J. J., T. L. Randell, M. Yamanaka, andK. L. Johnson. 1986. Pancreatic phospholipase A2: Isolation of the human gene and cDNAs from porcine pancreas and human lung.DNA 5:519–527.
Verkeij, H. M., A. J. Slotboom, andG. H. Dehaas. 1981. Structure and function of phospholipase A2.Rev. Physiol. Biochem. Pharmacol. 91:191–203.
Verkeij, H. M., J. Westerman, B. Sternby, andG. Dehaas. 1983. The complete primary structure of phospholipase A2 from human pancreas.Biochem. Biophys. Acta 747:93–99.
Hara, S., I. Kudo, H. Chang, K. Matsuta, T. Miyamoto, andK. Inoue. 1988. Amino acid composition and NH2-terminal amino acid sequence of human phospholipase A2 purified from rheumatoid synovial fluid.J. Biochem. 104:326–328.
Hara, S., I. Kudo, H. Chang, K. Matsuta, T. Miyamoto, andK. Inoue. 1989. Purification and characterisation of extracellular phospholipase A2 from human synovial fluid in rheumatoid arthritis.J. Biochem. (Tokyo) 105:395–399.
Lai, C., andK. Wada. 1988. Phospholipase A2 from human synovial fluid: Purification and structural homology to the placental enzyme.Biochem. Biophys. Res. Commun. 157:488–493.
Stefanski, E., W. Pruzanski, B. Sternby, andP. Vadas. 1986. Purification of a soluble phospholipase A2 from synovial fluid.J. Biochem. (Tokyo) 100:1297–1303.
Vadas, P., E. Stefanski, andW. Pruzanski. 1985. Characterisation of extracellular phospholipase A2 in rheumatoid synovial fluid.Life Sci. 36:579–578.
Seilhamer, J. J., W. Pruzanski, P. Vadas, S. Plant, J. A. Miller, J. Kloss, andL. K. Johnson. 1989. Cloning and recombinant expression of phospholipase A2 present in rheumatoid arthritic synovial fluid.J. Biol. Chem. 264:5335–5338.
Kramer, R. M., C. Hession, B. Johansen, C. Hession, B. Johansen, G. Hayes, P. McGray, E. P. Chow, R. Tizard, andR. B. Pepinsky. 1989. Structure and properties of a human non-pancreatic phospholipase A2.J. Biol. Chem. 254:5768–5775.
Vadas, P., andJ. B. Hay. 1980. The release of phospholipase A2 from aggregated platelets and stimulated macrophages of sheep.Life Sci. 26:1721–1729.
Takayama, K. I., S. Kudo, M. Hara, M. Murakami, K. Matsuta, T. Mijamoto, andK. Inoue. 1990. Monoclonal antibodies against human synovial phospholipase A2.Biochem. Biophys. Res. Commun. 167:1309–1315.
Suwa, Y., I. Kudo, A. Imaizumi, M. Okada, T. Kamimura, Y. Suzuki, H. W. Chong, S. Hora, andK. Inoue. 1990. Proteinaceous inhibitors of phospholipase A2 purified from inflammatory sites in rats.Proc. Natl. Acad. Sci. U.S.A. 87:2395–2399.
Buchler, M., A. Deller, P. Malfertheiner, H. O. Kleine, H. Wiedeck, W. Uhl, M. Samtner, H. Freiss, T. Nevalainen, andH. G. Beger. 1989. Serum phospholipase A2 in intensive care patients with peritonitis, multiple injury and necrotizing pancreatitis.Klin. Wochenschr. 67:217–221.
Hoffmann, G. E., andW. G. Guder. 1989. Serum phospholipase in regulatory and pathophysiological aspects.Klin. Wochenschr. 67:144–148.
Vadas, P., andW. Pruzanski. 1986. Role of secretory phospholipase A2 in the pathology of disease.Lab. Invest. 55:391–399.
Vadas, P., W. Pruzanski, andE. Stefanski. 1989. Phospholipase A2 in acute bacterial peritonitis in man.In Cell Activation and Signal Initiation. E. A. Dennis, T. Hunter, and M. Berridge, editors. Allan R. Liss, New York. 311–316.
Zalewski, A., S. Goldberg, andK. Maroko. 1988. The effects of phospholipase A2 inhibition on experimental infarct size, left ventricular hemodynamics and regional myocardial blood flow.Int. J. Cardiol. 21:247–257.
Chang, J., S. C. Gilman, andJ. A. Lewis. 1986. Interleukin-1 activates phospholipase A2 in rabbit chondrocytes: A possible signal for IL-1 action.J. Immunol. 136:1283–1287.
Godfrey, R. W., W. J. Johnson, andS. T. Hoffstein. 1988. Interleukin-1 stimulation of phospholipase activity in at synovial fibroblasts.Arthritis Rheum. 31:1421–1428.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Green, J.A., Smith, G.M., Buchta, R. et al. Circulating phospholipase A2 activity associated with sepsis and septic shock is indistinguishable from that associated with rheumatoid arthritis. Inflammation 15, 355–367 (1991). https://doi.org/10.1007/BF00917352
Issue Date:
DOI: https://doi.org/10.1007/BF00917352