Article Text
Abstract
Background As long-term treatment with antitumour necrosis factor (TNF) drugs becomes accepted practice, the risk assessment requires an understanding of anti-TNF long-term safety. Registry safety data in rheumatoid arthritis (RA) are available, but these patients may not be monitored as closely as patients in a clinical trial. Cross-indication safety reviews of available anti-TNF agents are limited.
Objective To analyse the long-term safety of adalimumab treatment.
Methods This analysis included 23 458 patients exposed to adalimumab in 71 global clinical trials in RA, juvenile idiopathic arthritis, ankylosing spondylitis (AS), psoriatic arthritis, psoriasis (Ps) and Crohn's disease (CD). Events per 100 patient-years were calculated using events reported after the first dose through 70 days after the last dose. Standardised incidence rates for malignancies were calculated using a National Cancer Institute database. Standardised death rates were calculated using WHO data.
Results The most frequently reported serious adverse events across indications were infections with greatest incidence in RA and CD trials. Overall malignancy rates for adalimumab-treated patients were as expected for the general population; the incidence of lymphoma was increased in patients with RA, but within the range expected in RA without anti-TNF therapy; non-melanoma skin cancer incidence was raised in RA, Ps and CD. In all indications, death rates were lower than, or equivalent to, those expected in the general population.
Conclusions Analysis of adverse events of interest through nearly 12 years of adalimumab exposure in clinical trials across indications demonstrated individual differences in rates by disease populations, no new safety signals and a safety profile consistent with known information about the anti-TNF class.
This is an open-access article distributed under the terms of the Creative Commons Attribution Non-commercial License, which permits use, distribution, and reproduction in any medium, provided the original work is properly cited, the use is non commercial and is otherwise in compliance with the license. See: http://creativecommons.org/licenses/by-nc/3.0/ and http://creativecommons.org/licenses/by-nc/3.0/legalcode
Statistics from Altmetric.com
Introduction
Adalimumab, a fully human monoclonal antibody targeted against tumour necrosis factor (TNF), is indicated for the treatment of six immune-mediated inflammatory diseases: rheumatoid arthritis (RA), juvenile idiopathic arthritis (JIA), ankylosing spondylitis (AS), psoriatic arthritis (PsA), psoriasis (Ps) and Crohn's disease (CD).
Because anti-TNF therapy suppresses the immune system,1 serious infections are the most frequently reported serious adverse events of interest across indications for the anti-TNF drug class.2 Given the role of TNF in mediating tumour growth,1 risk of malignancy with anti-TNF therapy has been a concern, although studies in RA have not shown a consistent safety signal.3 Complicating this risk assessment, there is substantial evidence that the chronic inflammation inherent in the conditions treated with anti-TNF therapy is itself associated with an increased potential for malignancy.2 ,4 ,5
Rates of adverse events in patients treated with anti-TNF agents can vary across therapeutic indications. Differences between populations (eg, disease-inherent risks, frequency of comorbidities and use of concomitant immunosuppressant drugs, including corticosteroids), may contribute to these differences.6
This analysis of the long-term safety profile of adalimumab through nearly 12 years of clinical trial exposure supplements registry safety data with well-monitored clinical trial data, highlights differences in adverse events between six patient groups, compares the risk of malignancy and mortality with the risk in the general population, examines temporal onset of adverse events and assesses two new events of interest—new onset/worsening of psoriasis and melanoma.
Patients and methods
Clinical trials
Data were derived from 71 adalimumab clinical trials, including randomised controlled trials, open-label trials and long-term extension studies conducted in Europe, North America, South America, Asia, Australia, New Zealand and South Africa, through 6 November 2010: 36 in RA, 3 in JIA, 4 in AS, 4 in PsA, 13 in Ps and 11 in CD. Safety data from adalimumab postmarketing surveillance were not included in this analysis to avoid limitations associated with voluntary reporting.7
Rates of serious adverse events of interest
Safety assessments included all adverse events that occurred after the first dose of adalimumab up to 70 days (five half-lives) after the last study dose. Serious adverse events were defined as fatal or immediately life-threatening; requiring inpatient hospitalisation or prolonging existing hospitalisation; resulting in persistent or significant disability/incapacity; congenital anomaly; or requiring medical or surgical intervention to prevent a serious outcome. Adverse events were coded using the Medical Dictionary for Regulatory Activities (MedDRA) preferred terms, MedDRA version 13.1 (http://www.meddramsso.com).
Serious adverse events of interest were identified using predetermined search criteria. All patients underwent medical review by company doctors. Rates are reported as events per 100 patient-years (PYs). Kaplan–Meier analyses were used to evaluate the time to first serious infectious event and the time to first malignancy/lymphoma/non-melanoma skin cancer (NMSC) for each indication.
Malignancy and mortality data for patients versus the general population
Standardised incidence rates (SIRs) were calculated as the ratio of observed to expected number of cancers; 95% CIs for SIRs were calculated assuming that observed cancers followed a Poisson distribution.8 The expected numbers of cancers, excluding NMSC, for SIR calculations were based on 5-year age-specific incidence rates from the National Cancer Institute (NCI) Surveillance Epidemiology and End Results (SEER) database, 1993–2001 (http://www.seer.cancer.gov). No similar database is available for Europe, Australia or Canada so an assumption was made that patients from these territories could be pooled with subjects from the USA. The NCI SEER database does not include NMSC; therefore, NMSC rates were based on 10-year age-specific incidence rates from a NCI survey of eight locations in the USA from 1977 to 1978.9
Standardised mortality rates were calculated as the ratio of observed deaths to expected deaths. Expected rates were estimated based on country-specific, age- and sex-matched population data from the WHO for 1997–2006 (http://www.who.int/whosis/database/mort/table1.cfm).
Results
Baseline characteristics and adalimumab exposure
Through 6 November 2010, adalimumab was administered to 23 458 patients, representing 36 730.5 PYs, a 23% increase in the number of adalimumab-treated patients and a 43% increase in PYs from a previous report.10 Baseline characteristics were representative of patients for each condition (table 1). The majority of adalimumab exposure was in RA studies (table 1), with over 65% of the total exposure and 60% of the total patients exposed, compared with 71% of the total exposure and 65% of the total patients treated in the previous report.10 The percentages of RA patients exposed >2 years (17.7%) and >5 years (11.7%) in this analysis were similar to the percentages in the 2009 Burmester dataset: 18.3% (2259/12 345) and 11.9% (1472/12 345), respectively.10
Baseline characteristics of adalimumab-treated patients and adalimumab exposure by therapeutic indication
Serious adverse events
Serious infection
Serious infectious events (SIEs) were the most frequently reported serious adverse events across all six therapeutic indications, with the greatest rates of SIEs seen in patients with RA or CD (table 2).
Incidence rates of serious adverse events of interest*
The most commonly reported SIEs (rates >0.2 events/100 PYs) were cellulitis (0.3/100 PYs) and pneumonia (0.7/100 PYs) in RA, appendicitis (0.5/100 PYs) and herpes zoster (0.3/100 PYs) in JIA, urinary tract infection (0.4/100 PYs) in PsA, cellulitis (0.3/100 PYs) in Ps, and gastrointestinal tract abscess (1.6/100 PYs), gastroenteritis (0.3/100 PYs) and pneumonia (0.4/100 PYs) in CD. No SIE rate exceeded 0.2/100 PYs in AS studies.
Risk of SIE was generally stable across time for all indications (figure 1). The majority of patients with SIEs (70%, 846/1208) across all indications continued adalimumab therapy. Approximately 32% of patients with RA and 28% of patients with CD with SIEs permanently discontinued treatment, compared with 18–24% of patients with JIA, AS, PsA and Ps. The most common SIEs leading to adalimumab discontinuation (>1% of patients with SIEs who discontinued adalimumab) were pneumonia (6.6%), bacterial arthritis (2.8%), gastrointestinal tract abscess (2.6%) and cellulitis (1.7%).
Time to first serious infection, by indication. AS, ankylosing spondylitis; CD, Crohn's disease; JIA, juvenile idiopathic arthritis; Ps, psoriasis; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
Active tuberculosis
Incidence rates for active tuberculosis (TB), excluding tuberculin test conversions with no evidence of active disease, were comparable to the previous analysis (0.29/100 PYs).10 No active TB was seen in patients with JIA and AS. Across all indications, the rate of active TB in adalimumab clinical trials was 0.2/100 PYs, including cases from RA clinical studies before implementation of a tuberculin test screening programme.11 Since latent TB infection screening and prophylaxis was implemented in 1998 and 1999, respectively, the rate has decreased from 1.5/100 PYs to 0.2/100 PYs.
Serious opportunistic infections
Twenty serious opportunistic infections, excluding TB and oral candidiasis, have been reported during adalimumab clinical trials (<0.1 events/100 PYs), 14 cases in patients with RA, four cases in patients with CD and two cases in JIA. The most common opportunistic infections were oesophageal candidiasis (n=3), and aspergillosis, Candida sepsis, coccidioidomycosis, cytomegalovirus infection, herpes zoster and nocardiosis (n=2 each). No serious opportunistic infections have been reported with adalimumab in AS, PsA or Ps clinical trials.
Serious demyelinating disorders, lupus-like syndrome, CHF
The incidence rates of serious demyelinating disorders, lupus-like syndrome and congestive heart failure (CHF) across all indications were ≤0.1 events/100 PYs, with the exception of CHF in patients with RA, which was 0.2/100 PYs (table 2). Four serious CHF events were reported for the first time in patients with Ps. No cases of progressive multifocal leucoencephalopathy have been reported in adalimumab clinical trials.
Serious new onset/worsening of psoriasis
The incidence of new onset/worsening of psoriasis, classified as psoriasis or pustular psoriasis, was very low, ≤0.1 events/100 PYs (table 2). No serious new onset/worsening of psoriasis events were reported in JIA studies.
Malignancies
Rates of malignancies reported in adalimumab clinical trials across all indications were 0.7 events/100 PYs for malignancies excluding lymphoma and NMSC, 0.1/100 PYs for lymphoma, and 0.2/100 PYs for NMSC; these rates were generally similar to previously reported rates.10 No malignancies were reported in JIA clinical trials with over 6 years of adalimumab exposure.
No cases of hepatosplenic T cell lymphoma were reported in any adalimumab study.
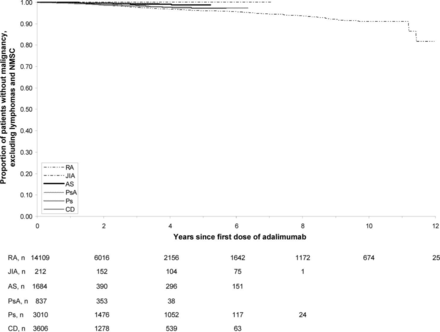
Kaplan–Meier analysis of time to first malignancy, for all malignancies excluding lymphoma and NMSC, did not show a marked difference between disease populations (figure 2). In all indications, the time to first lymphoma and time to first NMSC did not change with longer follow-up in this analysis. Risk of malignancy was generally low and remained stable over time.
Time to first malignancy, other than lymphoma or non-melanoma skin cancer (NMSC), by indication. AS, ankylosing spondylitis; CD, Crohn's disease; JIA, juvenile idiopathic arthritis; Ps, psoriasis; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
Malignancy rates compared with data from the general population
SIRs for all malignancies were based on 294 malignancies, including lymphomas but excluding NMSC, observed in adalimumab trials across indications (figure 3). Compared with age- and sex-matched populations, the observed number of malignancies in each disease population was similar to the expected number in the reference population.
Standardised incidence rates (SIR, (95% CI)) for all malignancies excluding NMSC, lymphomas and NMSC for RA, AS, PsA, Ps and CD. No malignancies were observed for juvenile idiopathic arthritis. aAll malignancies other than NMSC. bBased on data from 14 160 patients. AS, ankylosing spondylitis; CD, Crohn's disease; NMSC, non-melanoma skin cancer; Ps, psoriasis; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
SIRs for lymphomas (figure 3) were based on 29 events from RA, one from AS, two from PsA, one from Ps and two from CD trials (all non-Hodgkin's lymphoma except for six cases of Hodgkin's lymphoma in RA studies). The number of lymphomas observed in RA studies was significantly greater than expected compared with a US-based age- and sex-matched population (SIR=2.74; 95% CI 1.83 to 3.93).
SIRs for NMSC (figure 3) were based on 184 events (134 basal cell carcinoma (BCC), 43 squamous cell carcinoma (SCC) and six type unclassified)from RA, six (three BCC and three SCC) from AS, six (four BCC and two SCC) from PsA, 40 (26 BCC and 14 SCC) from Ps and 22 (14 BCC, six SCC, and two not classified) from CD trials. For NMSC, patients with RA, Ps and CD had SIRs (95% CIs) >1, indicating a higher number of observed NMSC cases than expected in the general population.
SIRs for melanomas were based on 15 observed events reported in RA and eight events in Ps studies. The observed number of melanoma events was raised in Ps, with a SIR (95% CI) of 4.37 (1.89 to 8.61). In patients with RA, the SIR (95% CI) of 1.5 (0.84 to 2.47), did not show a higher incidence relative to the general population.
Mortality rates compared with data from the general population
Deaths were reported in each adalimumab clinical programme except JIA. For subjects treated with adalimumab in RA, AS and Ps clinical studies, the observed number of deaths was less than expected in an age- and sex-matched population. In PsA and CD studies, the observed number of deaths was similar to the number expected in the reference population (figure 4).
Standardised death rates (SMR, (95% CI)) for all indications, RA, AS, PsA, Ps and CD. No deaths occurred in juvenile idiopathic arthritis. aNo deaths occurred among female patients with AS. AS, ankylosing spondylitis; CD, Crohn's disease; Ps, psoriasis; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
Discussion
This safety analysis represents the largest safety database published for an anti-TNF drug across indications, with up to 12 years of adalimumab exposure. In this analysis, infections were the most frequently reported serious adverse events across all therapeutic indications, consistent with previous safety analyses of adalimumab trials10 ,12,–,14 and other anti-TNF agents.15 The overall rate of SIE and the stable risk of first SIE throughout adalimumab exposure is consistent with findings previously reported.10 This differs somewhat from other risk estimates of anti-TNF therapy in RA, which suggest higher risk during the first 6–12 months of RA treatment.16 Possible explanations include population and methodological differences between clinical trials and observational registries.
The incidence rate of SIEs in patients with RA in this analysis is consistent with data on anti-TNF treatments from various RA registries, including the British Society for Rheumatology Biologics Register (BSRBR),6 the Spanish Society of Rheumatology drug registry (BIOBADASER),17 the German Rheumatoid Arthritis Observation of Biologic Therapy Registry (RABBIT)18 and the Swedish Biologics Register (ARTIS),19 which have reported rates ranging from 3.8 to 6.4 events/100 PYs in patients receiving anti-TNF therapy including etanercept and infliximab.6 ,15 ,18
Risk factors for SIEs with anti-TNF agents include disease-inherent and individual patient characteristics. Analysis of the anti-TNF cohort of the BSRBR6 as well as a population-based analysis from the Mayo Clinic20 showed that age, disease activity, comorbidities (eg, diabetes, chronic obstructive pulmonary disease) and baseline corticosteroid use were significant predictors of SIEs in patients with RA, regardless of treatment with disease-modifying antirheumatic drugs or biological agents. Corticosteroid use is associated with infections in patients with RA,21 and also in patients with CD, RA, AS, PsA and Ps, independently of anti-TNF use.22,–,24 In our analysis, lower SIE rates were seen in AS, PsA, Ps and JIA, where many of the aforementioned risk factors are not present. Nevertheless, clinicians must consider patient comorbidities, concomitant treatment and infection history when actively monitoring their patients for infection while treating with anti-TNF agents.
Other serious adverse events of interest occurred infrequently. The rates of active TB in adalimumab clinical trials across indications were similar to the rates previously reported.10 Opportunistic infections remain rare events. In the CORRONA registry for RA, a small risk of opportunistic infections was found with anti-TNF agents.25 Vigilant patient monitoring for opportunistic infections with anti-TNF agents, now including Legionella and Listeria infections,26 is recommended. Serious cases of demyelinating disorders, lupus-like syndrome, and CHF were uncommon, with low incidence rates generally remaining stable over time. Rates of serious CHF events remained as low as previously reported rates,10 even with expansion of search criteria for events classified as CHF (eg, pulmonary oedema, hepatic congestion). Of the four patients with Ps with serious CHF events, three had two or more risk factors for CHF, including hypertension, hyperlipidaemia, diabetes mellitus, myocardial ischaemia and obesity. The fourth patient developed pulmonary oedema secondary to a near drowning. The incidence of serious new onset/worsening of psoriasis was low. Drug-induced psoriasis is uncommon, but appears to be immune system related, and was reported with the anti- TNF agents etanercept, infliximab and adalimumab,27 as well as rituximab28 and anakinra,29 in patients treated for RA, AS, PsA, Ps and CD.
The overall rates of malignancy in adalimumab clinical trials were similar to rates expected in the reference population and consistent with rates previously published.10 ,30 Additionally, the time to onset of a first malignant event appeared stable across all indications, suggesting no increased risk over time with prolonged treatment.
Incidence rates of lymphoma in patients with RA were greater than those expected in age- and sex-matched populations without RA; however, data demonstrate that the risk of lymphoma is raised in patients with RA as a consequence of the disease.4 ,31 ,32 Chronic inflammatory activity is associated with lymphoma development, and patients with active RA are at greatest risk.4 Data from ARTIS showed that the risk of lymphoma in patients with RA receiving anti-TNF agents, although higher than in the general population, was the same as in patients never treated with anti-TNFs.31 Similarly, patients with RA in the Swedish Early Arthritis Register were found to have an inherent, nearly twofold increased risk for developing lymphomas during the first 10 years after diagnosis of RA, irrespective of treatment.31 The greater rate of lymphoma observed during adalimumab trials in patients with RA is within the range that might be expected in a similar RA population not treated with anti-TNF therapy.
Increased SIRs for NMSC in patients with RA, Ps and CD suggest there is a small but significant increase risk of NMSC with adalimumab compared with the risk observed in the NCI survey. NMSC SIRs can vary according to the reference database used,10 with SIRs generally greater using the NCI survey rather than other databases such as Arizona or Minnesota. This is probably because NCI demonstrated an increase in US skin cancer rates after the 1970s. Diagnosis of RA itself has been associated with a small risk of developing NMSC in a large national cohort of patients with RA.33 Data from observational studies and meta-analysis suggest an increased risk of NMSC with anti-TNF use30 ,34; however, the increased risk was associated with combination methotrexate and anti-TNF therapy and not with anti-TNF monotherapy.33 Patients with Ps and CD may also have an increased risk of NMSC. In Ps, use of high-dose psoralen and ultraviolet A light (PUVA) was associated with increased risk of NMSC.35 In a large retrospective cohort of patients with CD, recent use (≤90 days) and persistent use (>365 days) of adalimumab and infliximab were associated with increased odds of developing NMSC.36 However, it is not yet clear whether the underlying cause was related to detection bias, change in manifestation of NMSC or a true change in incidence.
Only the SIR in patients with Ps suggested an increased risk of melanoma with adalimumab. Most of these patients were exposed to other treatments such as ciclosporin, PUVA, retinoids and other immunosuppressants that have been associated with an increased risk of all skin malignancies.37 Additionally, the incidence of melanoma is increasing worldwide.38 Exposure to sunlight plus increased vigilance by dermatologists of these lesions might have contributed to these results. States of immune suppression have recently emerged as possible risk factors for melanoma.39 In RA, data have suggested an increased risk of melanoma associated with anti-TNF therapy.33 ,39 However, others have not found an increased risk of melanoma with anti-TNF agents at recommended doses for RA.40 ,41
The risk of mortality was not increased in adalimumab-treated patients compared with the reference population. Whether control of inflammation with adalimumab decreases certain mortality risks, such as cardiovascular disease42,–,44 and use of steroids,45 ,46 is yet to be determined. This hypothesis is supported by CORRONA RA registry data and a recently published meta-analysis showing that exposure to anti-TNF agents reduced non-infectious causes of mortality compared with patients not exposed, with reduction in cardiac mortality appearing to be the major reason for this reduced risk.42 ,47
Several limitations exist in the interpretation of the findings of this analysis. Protocol-specified patient selection probably resulted in study populations with fewer comorbidities than the wider general patient population. Comparisons with other treatments could not be determined owing to lack of a control group in the long-term open-label periods. The reference population for malignancy SIRs was a US-based population, which may limit the generalisability of these global clinical trial results. Finally, patients in the adalimumab clinical trial programme were closely monitored at regular scheduled visits, which might have resulted in detection bias for adverse events.
The adalimumab safety data presented here provide important additional support for the long-term safety of adalimumab in six different immune-mediated inflammatory diseases, highlighting important differences among patient populations, and demonstrating stability of incidence rates over time with no new safety signals.
Acknowledgments
The authors thank Kathleen V Kastenholz, PharmD, MS, of Abbott Laboratories, for medical writing and editing support in the development and revision of this manuscript. The authors also thank Joseph Beason, of Abbott Laboratories, for his statistical support.
References
Supplementary materials
Supplementary Data
This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Files in this Data Supplement:
- Web Only Data - This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
- Web Only Data - This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
- Web Only Data - This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
- Web Only Data - This web only file has been produced by the BMJ Publishing Group from an electronic file supplied by the author(s) and has not been edited for content.
Footnotes
-
Funding Abbott Laboratories sponsored the clinical trials and contributed to their design and data analysis.
-
Competing interests GRB has served as a consultant to Abbott Laboratories, Essex/Schering-Plough, Novartis and Roche and has received grants and honoraria from Abbott, Essex/Schering-Plough, Novartis, Roche, and Wyeth. RP has served as a consultant to Abbott Laboratories, AstraZeneca, Bristol-Myers Squibb, Centocor, Elan, Ferring, GlaxoSmithKline, Procter and Gamble, Schering-Plough, Shire and UCB and has received grants from Abbott, Axcan, Bristol-Myers Squibb, Centocor, Elan, Millennium and Procter & Gamble and honoraria from Abbott Laboratories, Astra Zeneca, Byk Solvay, Centocor, Elan, Janssen, Procter and Gamble, Prometheus, Schering-Plough and Shire. KBG has received honoraria from and served as a consultant to Abbott Laboratories, Amgen, Centocor, Galderma, Pfizer, Merck and Lilly and has received grants from Abbott, Amgen, Centocor and Celgene. MJM and AL are employees of Abbott Laboratories and may hold stock or stock options.
-
Ethics approval National, regional, or investigative site ethics committees/institutional review boards, as appropriate.
-
Provenance and peer review Not commissioned; externally peer reviewed.
-
Correction notice This article has been corrected since it was published Online First. The unlocked logo has been added and fi gure 3 has been corrected.